Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each
Question:
Each of the following compounds has a single 1H NMR peak. Approximately where would you expect each compound toabsorb?
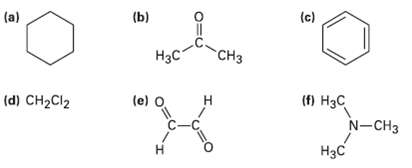
Transcribed Image Text:
(c) (b) (a) Нас "СHз (d) CH2CI2 (e) O (п Нас N-CHз н C-C Нзс н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Compound a C6H12 b CH3COCH 3 c C6H6 d CHCl ...View the full answer

Answered By

Vineesh kumar V
To work in your esteemed organization where I can prove my expertise and work towards the growth of the organization
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds has been prepared from p-nitroaniline. Outline a reasonable series of steps leading to each one. (a) p-Nitrobenzonitrile (d) 3, 5-Dibromoaniline (b) 3, 4,...
-
Each of the following compounds has been prepared from o-anisidine (o-methoxyaniline). Outline a series of steps leading to each one. (a) o-Bromoanisole (d) 3-Fluoro-4-methoxybenzonitrile (b)...
-
According to Thomson Financial, last year the majority of companies reporting profits had beaten estimates. A sample of 162 companies showed that 114 beat estimates, 29 matched estimates, and 19 fell...
-
How would you characterize the nature of competition in the restaurant industry? Are there submarkets with distinct competitive pressures? Are there important substitutes that constrain pricing?...
-
By how much does adiposity (a measure of body fat) differ between adult women who maintain a regular sleep schedule versus women whose sleep schedule fluctuates by 90 minutes or more? For each study,...
-
The data in Table 19.8 are from Gamage et al. (2007). These data are for 36 months of medical-care payments, from January 2001 through December 2003, inclusive. These are payments for medical-care...
-
Dillon Products manufactures various machined parts to customer specifications. The company uses a job-order costing system and applies overhead cost to jobs on the basis of machine-hours. At the...
-
Analysis of short selling strategy and its role played in Tesla short squeeze Discussion of why short squeeze is not going to happen to Tesla in 2021?
-
Public Company acquired 85% of the common stock of Sample Company January 1, year one, for $510,000 The consideration given was proportional to Sample's fair value. On that date, Sample had the...
-
How many absorptions would you expect S)-malate, an intermediate in carbohydrate metabolism, to have in its NMR spectrum?Explain. (S)-Malate
-
Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect each toabsorb. CH-CH C
-
Discuss and contrast the various roles in data management. Clearly indicate the key activities and skills required. Discuss which job profiles can be combined.
-
what ways does the study of phenotypes inform our understanding of complex biological phenomena, including disease susceptibility, ecosystem dynamics, and evolutionary processes, providing insights...
-
The binomial 2-49 has y - 7 as one of its factors. What is the other factor of the binomial?
-
Use the Zero Property Rule to find the solution (s). x^(2)+9x+14=0
-
Can you elaborate on the concept of phenotypic integration, referring to the coordinated variation and functional relationships among multiple traits within an organism, and how does phenotypic...
-
Choose a marketing/ business you'd like to solve ? How would Isolve the issue of online vs in person retail shopping? how do I entice shoppers to in person retail shopping with the convenience of...
-
The data set in Table 2.9 consists of the profits/losses (in millions of dollars) for a random sample of 20 companies. a. Draw a histogram of the company profits/losses for this sample of companies...
-
What is a manufacturing system?
-
Calculate the osmotic pressure of a solution containing 24.6 g of glycerin (C 3 H 8 O 3 ) in 250.0 mL of solution at 298 K.
-
Unlike ethylene glycol, propylene glycol (propane-1,2-diol) is nontoxic because it oxidizes to a common metabolic intermediate. Give the structures of the biological oxidation products of propylene...
-
Predict the major products of the following reactions. (a) Ethyl tosylate + potassium tert butoxide (b) Isobutyl tosylate + NaI (c) (R) 2 hexyl tosylate + NaCN (d) The tosylate of cyclohexlmethanol +...
-
Complete the following conversion table. 1700 1640 1600 400 T(cm ) A() 2.50 3.03 3.33 4.55 4000 25.0
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....
-
The cash flow for the firm's project is -$40 million in year 0 and $19 million in years 1-4. After year 4, the FCF is expected to grow at a constant rate of 0.020. The firm's discount rate is 0.068....

Study smarter with the SolutionInn App


