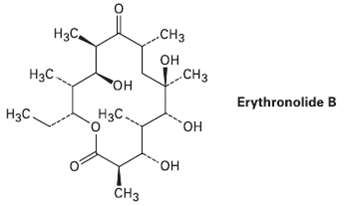
Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. How many chirality centers does Erythronolide
Question:
Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. How many chirality centers does Erythronolide Bhave?
Transcribed Image Text:
Нас, .CНз он сHз Нас. CH3 но. Erythronolide B Нзс Нас. но, но. CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
H3C HC H3C OH H3C C...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many chirality centers are contained in (a) The aldotetrose (b) The ketopentose just given? (c) How many stereoisomers would you expect from each general structure?
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
How many stereogenic centers are present in squalene? In lanosterol?
-
An automobile is traveling at 60.0 km/h. Its tires have a radius of 33.0 cm. (a) Find the angular speed of the tires (in rad/s). (b) Find the angular displacement of the tires in 30.0 s. (c) Find the...
-
Would buoyancy occur in the absence of weight? Discuss the buoyancy that would or wouldn't occur in the International Space Station.
-
Use the moment-distribution method to determine the moment at each joint of the frame. The supports at A and C are pinned and the joints at B and D are fixed connected. Assume that E is constant and...
-
Merit increases require a single performance number, while most incentive plans have multiple and varying performance measures. How can the PM system meet both needs?
-
Carolinas Golf School, Inc., completed the following transactions during October, 2012: Oct 1 Prepaid insurance for October through December, $900. 4 Performed services (gave golf lessons) on...
-
Grove Company has budgeted the following credit sales for the last six months of the current year. GROVE COMPANY BUDGETED CREDIT SALES FOR LAST SIX MONTHS OF CURRENT YEAR July $ 3 0 0 , 0 0 0 October...
-
Bonivo Inc. manufactures computers from commodity components to client specifications. The company has historically tracked only the cost of components to computers, and computer selling prices, or...
-
Which of the following objects arc chiral? (a) A basketball (b) A fork (c) A wine glass (d) A golf club (e) A monkey wrench (f) A snowflake
-
Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the other S
-
An investor tells you that in her estimation there is a 60% probability that the Dow Jones Industrial Averages index will increase tomorrow. a. Which approach was used to produce this figure? b....
-
A 95 kg quarterback accelerates a 0.42 kg ball from rest to 24 m/s in 0.083 s. Part A What is the specific power for this toss? Express your answer in watts per kilogram. SPLoss ? W/kg Submit Request...
-
Create a Python class representing a circle. Include methods to calculate its area and circumference.
-
If the value of the unlevered firm with taxes is 171m and the levered firm has 111m of debt, what the value of the levered firm assuming a tax rate of 40%. Assume the firms are identical in every way...
-
Find the value of Caspian Sea Corp. assuming corporate taxes: ke=9.7% kd=5% It currently has no debt EBIT=$2m T=40% Show your answer in millions notation to two decimal places. (i.e., $500,000 would...
-
A stock exchange trader contacts you and explains that he is looking to enter the cryptocurrency market with his own product that is named Z1. The coins currently have little to no value other than...
-
Assume that the water utility, an enterprise fund, was billed $25,000 for data processing work during the year ended June 30, 20X7. What account in the enterprise fund should be debited to record...
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
What is the pressure of 1.78 g of nitrogen gas confined to a volume of 0.118 L at 25 C? a) 13.2 atm b) 369 atm c) 1.10 atm d) 26.3 atm
-
Outline a synthesis of each of the following compounds from the indicated strating material and any other reagents. (a) N-(sec-buty1) -N-ethylaniline from chlorobenxene (b) (c) (CH,),C from phenol NH...
-
(a) Could ten-butylamine be prepared by the Gabriel synthesis? If so, write out the synthe-sis. If not, explain why. (b) Propose a synthesis of tert-butylamine by another route.
-
Write a curved-arrow mechanism for the following reactions. Ethyl isocyanate with ethylamine to yield N,N'-diethylurea
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1: 1. 73,000 shares of $4 par common stock. 2. 7,000 shares of $90 par, 6 percent, noncumulative preferred stock....
-
Teresa is a 32-year-old woman in your practice who frequently misses her appointments and at other times shows up without an appointment, often in crisis. She currently uses alcohol and tobacco, and...
-
On January 1, Year 1, Grapefruit Corporation purchased a machine for $41,300,000. Grapefruit's management expects to use the machine for 26,000 hours over the next six years. The estimated residual...

Study smarter with the SolutionInn App


