Estimate the liquid diffusivity of acetic acid at 25oC in a dilute solution of:(a) Benzene,(b) Acetone,(c) Ethyl
Question:
Estimate the liquid diffusivity of acetic acid at 25oC in a dilute solution of:(a) Benzene,(b) Acetone,(c) Ethyl acetate, and(d) Water by an appropriate method. Compare the estimated values with the following experimentalvalues: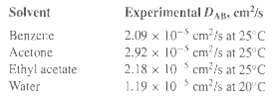
Transcribed Image Text:
Experimental DA, em/s 2.09 x 10- cm /s at 25 C 2.92 x 10 cm'/s at 25 C 2.18 x 10 cm/s at 25°C 1.19 x 10 cm'is at 20'C Solvent Вептете Acetone Ethyl acetate Water
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
For benzene acetone and ethyl acetate Eq 342 of HaydukMinhas applies From Table 35 the para...View the full answer

Answered By

User l_695272
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Estimate the diffusivity of carbon tetrachloride at 25oC in a dilute solution of:(a) Methanol,(b) Ethanol,(c) Benzene, and(d) n-Hexane by the method of Wilke-Chang and Hayduk-Minhas. Compare the...
-
Estimate the liquid diffusivity of benzene (A) In formic acid (B) At 25C and infinite dilution. Compare the estimated value to that of Example 3.6 for formic acid at infinite dilution in benzene.
-
At 25oC in a closed system, ammonium hydrogen sulfide exists as the following equilibrium: a. When a sample of pure NH4HS(s) is placed in an evacuated reaction vessel and allowed to come to...
-
Which of the following domains are closed and which are bounded? (a) {(x, y) R: x + y 1} (b) {(x, y) = R: x + y <1} (c) {(x, y) = R: x 0} (d) {(x, y) R: x > 0, y > 0} (e) {(x, y) = R: 1 x 4,5 y 10}...
-
In 2012, Westlake Ltd. had the following transactions related to the purchase of property. Assume all transactions Feb. 7 Purchased real estate for $550,000, paying $150,000 cash and signing a...
-
In 2020, Carson is claimed as a dependent on his parents tax return. His parents report taxable income of $200,000 (married filing jointly). Carsons parents provided most of his support. What is...
-
A mutual fund has provided investment yield rates for five consecutive years as follows: Determine \(r_{1}\) and \(r_{2}\), the lag 1 and lag 2 autocorrelation coefficients. Determine \(r_{1}\) and...
-
The following information was taken from the records of Liberty Tool Manufacturing Company for its production of jackhammers in the month of July: Required: 1. Calculate the materials price and...
-
Gold Inc invests $10,000 today in a mutual fund. Gold anticipates leaving this fund alone for 12 years. $ The fund is increased each year-end by specified compound interest rates as follows: years 1...
-
What discount rate is the Powerball lottery company using when deciding to pay a lump sum of $327.8m to each of the 3 winning ticket holders?
-
For the mixture of Example 3.3, estimate the diffusion coefficient if the pressure is increased to 100 atm using the method of Takahashi.
-
Water in an open dish exposed to dry air at 25oC is found to vaporize at a constant rate of 0.04 g/h-cm2. Assuming the water surface to be at the wet-bulb temperature of ll.0C, calculate the...
-
Rejecting the null hypothesis? If F cv = 3.259 and F = 3.259, was H 0 rejected?
-
What should be considered before including alternative media in an IMC campaign?
-
Define experiential marketing and its use.
-
The U.K. Met Office site https://www.metoffice.gov.uk/ keeps long-term weather records-temperature, rainfall, and so on-for a range of stations in Great Britain and Northern Ireland. Monthly rainfall...
-
What are the pros and cons of using alternative media?
-
Use fft () to compute the Fourier coefficients for the temperature series on a whole number of years, identify and remove the frequencies that are seasonal, average the power-spectrum values in...
-
Amy Stewart has always been interested in stocks. She has decided to invest $2,000 once every year into an equity mutual fund that is expected to produce a return of 6% a year for the foreseeable...
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Identify the three characteristics of an effective benchmark for an SAA analysis.
-
Can a column be operated at total reflux? How?
-
A column at 101 kPa is to separate 30 kg/h of a bubble-point solution of benzene and toluene containing 0.6 mass-fraction toluene into an overhead product of 0.97 mass-fraction benzene and a bottoms...
-
In the McCabeThiele method, are the stages stepped off from the top down or the bottom up? In either case, when is it best, during the stepping, to switch from one operating line to the other? Why?
-
Suppose the North Carolina University system tests the idea that computerization of statistics classes increases student performance in the courses. If the computers are found to have a beneficial...
-
M. Handy is a self-employed painter who earned $32,400 last year. Her FICA tax rate is 15.3% of her earnings. How much FICA tax did she pay?
-
Using the same iThe Shouldice Hospital specializes in hernia operations. They have a special procedure that not only has a much higher rate of long-term success than other hospitals, but also allows...

Study smarter with the SolutionInn App


