For a given value of [HL]org in Equation 22-13, over what pH range (how many pH units)
Question:
For a given value of [HL]org in Equation 22-13, over what pH range (how many pH units) will D change from 0.01 to 100 if n = 2?
Equation 22-13
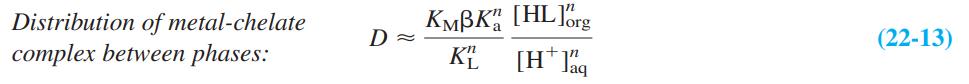
Transcribed Image Text:
Distribution of metal-chelate complex between phases: KMBK [HLErg K“ [H* ig D (22-13)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
D CH n where C K M n a HL n o...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Show that the number of different electron states possible for a given value of n is 2n2.
-
(a) For a given value of the principal quantum number n, how many values of the orbital quantum number are possible? (b) For a given value of how many values of the orbital magnetic quantum number m...
-
For a given value of the principal quantum number, n, how do the energies of the s, p, d, and f sub-shells vary for? (a) Hydrogen, (b) A many-electron atom
-
Why do countries respond differently in terms of flexibility and worklife balance to what seem similar economic pressures?
-
You are provided with the following information for Barton Inc. Barton Inc. uses the periodic method of accounting for its inventory transactions. March 1 Beginning inventory 2,000 liters at a cost...
-
Lew Pherson and Vera Collier are friends who are employed by different CPA firms. One day during lunch they are discussing the importance of internal control in determining the amount of audit...
-
What is the purpose of an insurance policy?
-
Varmit-B-Gone is a pest control service that operates in a suburban neighborhood. The company attempts to make service calls at least once a month to all homes that subscribe to its service. It makes...
-
Andrew Ghez is the new chief financial officer ( CFO ) for Millcreek Company. Andrew has a long career as a financial accountant. In his new role as CFO, he will be supervising the work of both the...
-
The rigid bar is pinned at A and supported by two aluminum rods, each having a diameter of 1 in. and a modulus of elasticity E al = 10(10 3 ) ksi. If the bar is initially vertical, determine the...
-
Butanoic acid has a partition coefficient of 3.0 (favoring benzene) when distributed between water and benzene. Find the formal concentration of butanoic acid in each phase when 100 mL of 0.10 M...
-
For the extraction of Cu 2+ by dithizone in CCl 4 , KL= 1.1 10 4 , K M = 7 10 4 , K a = 3 10 - 5 , = 5 10 22 , and n = 2. (a) Calculate the distribution coefficient for extraction of 0.1 M Cu 2+...
-
Which nucleophile, ;N(C 2 H 5 ) 3 or ;P(C 2 H 5 ) 3 , reacts most rapidly with methyl iodide in ethanol solvent? Explain, and give the product formed in each case.
-
Consider a one-period exchange economy with two agents \(i \in\{1,2\}\), two possible states of the world \(\left\{\omega_{1}, \omega_{2} ight\}\) and two consumption goods. Suppose that...
-
A: When an M&M is randomly selected from the 100 M&Ms listed in Data Set 20, it is one of the 8 yellow M&Ms. B: When a second different M&M is randomly selected from those listed in Data Set 20, it...
-
If 1 of the 839 challenges is randomly selected, find the probability of getting a challenge that was rejected. Use the following results from the 839 player challenges to referee calls in the first...
-
Find the probability of randomly selecting 2 different challenges and finding that they were both accepted challenges made by female players. Use the following results from the 839 player challenges...
-
Find the probability of randomly selecting 1 of the 839 challenges and getting a challenge that was accepted and was made by a female player. Use the following results from the 839 player challenges...
-
Using the Laplace transform and showing the details of your work, solve the IVP: y' 1 = 4y 1 + y 2 , y 2 = -y 1 + 2y 2 , y 1 (0) = 3, y 2 (0) = 1
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
Quartz crystal microbalance. The area of the gold electrodes on the quartz crystal microbalance at the opening of Chapter 2 is 3.3 mm 2 . One gold electrode is covered with DNA at a surface density...
-
(a) Figure 1-1 shows a peak O 3 concentration of 19 mPa in the stratosphere. Figure 1-3 shows a peak concentration of O 3 of 39 ppb at ground level at one particular location. To compare these...
-
(a) How much of the primary standard benzoic acid (FM 122.12, density = 1.27 g/mL) should you weigh out to obtain a 100.0 mM aqueous solution in a volume of 250 mL? (b) What apparent mass in air will...
-
Annuity Payments Go to www.fcfcorp.com/onlinecalc.htm. Use the calculator to solve this problem. If you have $1,500,000 when you retire and want to withdraw an equal amount for the next 30 years, how...
-
Calculating Future Values Go to www.dinkytown.net and follow the Savings Calculator link. If you currently have $10,000 and invest this money at 9 percent, how much will you have in 30 years? Assume...
-
In 2023, Amanda and Jaxon Stuart have a daughter who is 1 year old. The Stuarts are full-time students and are both 23 years old. Their only sources of income are gains from stock they held for three...

Study smarter with the SolutionInn App


