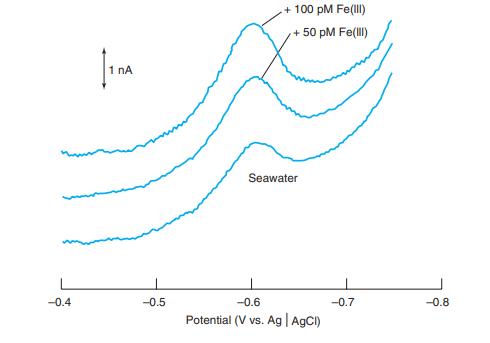
From the two standard additions of 50 pm Fe(III) in Figure 16-23, find the concentration of Fe(III)
Question:
From the two standard additions of 50 pm Fe(III) in Figure 16-23, find the concentration of Fe(III) in the seawater. Estimate where the baseline should be drawn for each trace and measure the peak height from the baseline. Consider the volume to be constant for all three solutions.
Figure 16-23
Transcribed Image Text:
+ 100 pM Fe(ll) +50 pM Fe(lI) 1 nA Seawater --0.4 -0.5 -0.6 --0.7 -0.8 Potential (V vs. Ag AgCI)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Cells B13 and B18 of the spreadsheet tell us that F...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The figure below shows a series of standard additions of Cu 2+ to acidified tap water measured by anodic stripping voltammetry at an iridium electrode. The unknown and all standard additions were...
-
Standard addition. A particular CO2 compound electrode like the one in Figure 14-28 obeys the equation E = constant - [RT (ln 10)/2F] log[CO2], where R is the gas constant, T is temperature (303.15...
-
The method of standard additions was used to determine nitrite in a soil sample. A 1.00-mL portion of the sample was mixed with 24.00 mL of a colorimetric reagent, and the nitrite was converted to a...
-
A space-filling curve is a continuous curve in the unit square that passes through every point. Write a recursive Turt le client that produces these recursive patterns, which approach a space-filling...
-
Benedict Company incurred the following costs. 1. Sales tax on factory machinery purchased ........... $ 5,000 2. Painting of and lettering on truck immediately upon purchase ... 700 3. Installation...
-
A model for a hemodialyser with simulation of the patient-artificial-kidney system: a case-study problem. A useful case study is the paper by Ramachandran and Mashelkar (1980), where a mesoscopic...
-
Find the power spectral density of a doubly stochastic Poisson impulse process having a rate process \(\Lambda(t)\) described by \[ \Lambda(t)=\lambda_{0}[1+\cos (2 \pi \bar{v} t+\Phi)] \] where...
-
Pendergrass Company hires an accounting intern who says that intangible assets should always be amortized over their legal lives. Is the intern correct? Explain.
-
Let's take a step back from the forget/borrow/learn framework. Many of you raised the issue in an earlier thread regarding the relationship between the old co and new co. It is unrealistic to expect...
-
Oregon Corporation has filed a voluntary petition to reorganize under Chapter 11 of the Bankruptcy Reform Act. Its creditors are considering an attempt to force liquidation. The company currently...
-
The standard free energy change for the formation of H 2 (g) + O 2 (g) from H 2 O is G = + 237.13 kJ. The reactions are Calculate the standard voltage (E) needed to decompose water into its elements...
-
Peak current (Ip) and scan rate (I p ) are listed for cyclic voltammetry (Fe(II) Fe(III)) of a water-soluble ferrocene derivative in 0.1 M NaCl. If a graph of I p versus v gives a straight line,...
-
A citizen decides to campaign for the election of a candidate for city council. Her goal is to generate at least 210 votes by a combination of door-to-door canvassing, letter writing, and phone...
-
In the story "A Devoted Son" by Anita Desai, Rakesh, the son, touches his father Varma's feet. What is the significance of this, and how does it change as the story progresses?
-
9) When a beam of light originally traveling in air, enters a piece of glass NORMALLY, its wavelength A) increases by a factor B) is reduced by a factor C) is unaffected.
-
After being struck by a bowling ball, a 1.4 kg bowling pin sliding to the right at 3.7 m/s collides head-on with another 1.4 kg bowling pin initially at rest. Find the final velocity of the second...
-
13. The Apple stock's price is $112.92 on 8/1/15 and becomes $110.30 on 9/1/15. In August, Apple gives a dividend of $0.52 per share. What is the holding period monthly return for Apple in August? A....
-
A block of wood ( with dimensions of 6 0 , 3 0 , and 4 cm ) floats on water with 8 0 % of its volume below the surface of water. Density of water is 1 0 0 0 kg / m 3 . a . Find the buoyant force...
-
In the late 1970s, Sony, a manufacturer of video cassette players, and Columbia Pictures, a movie studio, played a game involving the choice of technology. Each firm had to decide whether to use the...
-
If there is an unrealized holding gain on available-for-sale investments, it is reported as?
-
In the series SiF4, PF3, and SF2, estimate the F-X-F bond angle in each case and explain your rationale.
-
(a) If the valence atomic orbitals of an atom are sp hybridized, how many unhybridized p orbitals remain in the valence shell? How many bonds can the atom form? (b) Imagine that you could hold two...
-
(a) Draw Lewis structures for ethane (C2H6), ethylene (C2H4), and acetylene (C2H2). (b) What is the hybridization of the carbon atoms in each molecule? (c) Predict which molecules, if any, are...
-
What is thread affinity, and how does it influence performance in multi-core systems? What tools or techniques are available for tuning thread affinity ?
-
Management of Mittel Company wants to reduce the elapsed time from when a customer places an order to when it is shipped. It provided the following data for a recent quarter: Inspection time Wait...
-
For a language L over alphabet , we define L = {xz *y * with |x| = |y| = |2| such that ryz L}. For example, if L = {a, to, cat, math, solve, theory}, then L = {ct,thry}. Prove that if L is regular,...

Study smarter with the SolutionInn App


