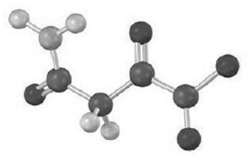
From what amino acid is the following ?-keto acid derived?
Question:
From what amino acid is the following ?-keto acid derived?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
As in the previous problem redraw the aketo aci...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The isoelectric point of an amino acid is the pH at which the molecule has no net charge. For glycine, that would be the pH at which virtually all glycine molecules are in the form +H3NCH2CO2-. If we...
-
Amino acid compositions can be determined by heating a protein in 6 M HCl and running the hydrolysate through an ion-exchange column. If you were going to do an amino acid sequencing experiment, why...
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
Bookstores International, a bookstore chain, has been quite successful over the past few decades and is now in expansion mode. It typically approaches opening a new bookstore by first determining the...
-
What are the primary disadvantages and advantages of a reverse merger strategy?
-
A 750 g disk and a 760 g ring, both 15 cm in diameter, are rolling along a horizontal surface at 1.5 m/s when they encounter a 15 slope. How far up the slope does each travel before rolling back down?
-
Upon reviewing recent use of conference rooms at an engineering consulting firm, an industrial engineer determined the following probability distribution for the number of requests for a conference...
-
Assume a condition in which the economy is strong, with relatively high employment. For one reason or another, the money supply is increasing at a high rate and there is little evidence of money...
-
The weekly demand of fresh oranges, as measured by number of oranges, for a household depends on the price of orange (in dollars) and can be approximated by q(p) = 20-p. Suppose that the supply of...
-
You are assessing internal control in the audit of the payroll and personnel cycle for Rogers Products Company, a manufacturing company specializing in assembling computer parts. Rogers employs...
-
What -keto acid is formed on transamination of leucine?
-
Identify the amino acid that is a catabolic precursor of each of the following ?-keto acids: (b) (a)
-
Comment on the management of confict with reference to styles of confict management, the use of superordinate goals, and formal negotiations.
-
The current spot exchange rate for the New Zealand Dollar per Euro (NZD/EUR) is 1.6129. The one-year risk-free rates are 3.5% for the New Zealand dollar and 1.2% for the Euro. According to CIP, the...
-
In the spring of year 2000, Silicon Valley is considering the issue of a convertible bond. The issue would consist of 20 year convertible bonds that would sell at a price of $1,000 per bond. The bond...
-
Review the following sales information: January sales are $ 60,000 February sales are $ 70,000 80% of sales are made on a cash basis 20% of sales are collected after 30 days How much cash will the...
-
Section 1: Current Marketing Situation Situational Analysis including Threats and Opportunities Analysis and Objectives and Issues This section describes the situation surrounding your business or...
-
In a given population of men and women, 20% of the men are married and 30% of the women are married. What percentage of the adult population is married? Assume that, in this particular population,...
-
Use the defining integral to find the Fourier transform of \(f(t)=A[u(t+1)-u(t-2)]\).
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
Redo Problem 13.28 using Aspen Plus. Problem 13.28 A gas mixture containing equimolar quantities of carbon dioxide and hydrogen is to be reformed by passing it over a catalyst. The pressure in the...
-
Write an equation for the reaction of ammonia (:NH3) with hydrogen chloride (HCl). Use curved arrows to track electron movement, and identify the acid, base, conjugate acid, and conjugate base.
-
As noted in Problem 4.7, hydrogen cyanide (HCN) has a pKa of 9.1. Is cyanide ion (CN-) a stronger base or a weaker base than hydroxide ion (HO-)?
-
Write an equation for proton transfer from hydrogen chloride to tert-butyl alcohol. Use curved arrows to track electron movement, and identify the acid, base, conjugate acid, and conjugate base.
-
Hi, could someone please help me with this. I need the letter to be nearly two hundred words. This is the assignment below: You are an architect who is working for Dream Homes Company in Toronto....
-
Discussion Board Post - Read the speech example from the textbook below. Then answer the questions in blood at the bottom. "As a veterinarian and owner of a small-animal practice, you work closely...
-
The Essentials of Technical Communication (Tebeaux & Dragga), Oxford U. Press, Third Edition - I. Please explain how a proposal is an argumentative or persuasive piece of writing -- and the four key...

Study smarter with the SolutionInn App


