From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each
Question:
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.)
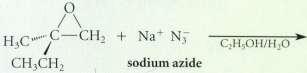
Transcribed Image Text:
C,H OH/H,O CH CH2 sodium azide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The strategy in this problem is to let the OH group originate fro...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds is an aromatic compound bearing a substituent that we did not discuss in this chapter. Using the principles that we discussed in this chapter, predict the major...
-
Can the following compound be prepared via a Williamson ether synthesis? Explain your answer.
-
The portfolio of stock that comprises the ASX200 index is currently worth $5000. The continuously compounded interest rates on Australian government bonds is 1.5% per annum for each of the next five...
-
Assume initially that Demand and supply for premium coffees (one pound-bags) are in equilibrium. Now assume Starbucks introduces the world to premium blends so Demand rises substantially. Describes...
-
Heather Oak is trying to prepare a personal budget and has identified the following list of monthly costs. Identify each cost as fixed, variable, or mixed. Indicate a possible cost driver for any...
-
Consider the sample space \(S=\{y y, y n, n y, n n\}\) in Example 2.2. Suppose that the subset of outcomes for which at least one camera conforms is denoted as \(E_{1}\). Then, \[ E_{1}=\{y y, y n, n...
-
Shop Rite Services is ready to prepare its financial statements for the year ended December 31, 2012. The following information can be determined by analyzing the accounts: 1. On August 1, 2012, Shop...
-
Fairfield Company's raw materials inventory transactions for the most recent month are summarized here: Note: Assume, purchase of raw materials is on account. Beginning raw materials Purchases of raw...
-
Albus was opening a landscaping business and planned on hiring 2 assistants. He advertised in the local newspaper and on an internet job site to find the right employees. Of the applicants, Albus...
-
What products are formed when gach of the following ethers reacts with concenffated aqueous HI? 2-ethoxy-2,3-dimethylbutane
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
Best Drugs is a distributor of pharmaceutical products. Its ABC system has five activities: Activity Area Cost Driver Rate in 2017 1. Order processing............................$42 per order 2....
-
When discussing the PCI DSS standard in the materials, it may be hard to think that there are a lot of industries which are affected by this standard. What industries can you think of that have had...
-
Explain the anomalies detected within the original 10-K. Consider the following questions to help guide your response: Are sales increasing? Are accounts receivable decreasing? Are write-offs...
-
Actual value is at the highest dotted line, right under is the earned value and at the lowest is the actual cost. a. At the time of the dotted line is the project over budget, on budget, or under...
-
For 2021, calculate ROCE (return on common equity). Is there any important difference with respect to ROE for the same year? Please explain. (Hint: assume that preferred equity does not grant any...
-
According to Modigliani and miller, the firm's choice of dividend policy is irrelevant. Empirically, firms increase dividends much more frequently than they cut them. Discuss these theories in...
-
The following is an accounts receivable ageing schedule for Cain Ltd. At 31 December 2016 the unadjusted balance in Allowance for Doubtful Debts is a credit of $10 000. Required (a) Journalise and...
-
Draw the appropriate control flow graph of the given pseudocode.Make sure to only use one number for blocks of code which are all sequential and when the first line is executed, all of those lines...
-
Provide a reaction mechanism for step 1, formation of the imine, in Eq. 23.22. EtNH + H3C-C-CH3 acetone -HO [HC- NEt H3C-C-CH3 an imine (not isolated) H, Pt 30 psi EtOH NHET H3C-CH-CH3 (23.22)...
-
Provide reaction mechanisms for the following equations. (a) Eq. 23.17 (b) Eq. 23.18 (c) Suggest a reason why the reaction in Eq. 23.18 stops after two additions, and a third doesnt occur in high...
-
Using their solubilities in acidic or basic solution, design a separation of p-chlorobenzoic acid, p-chloroaniline, and p-chlorotoluene from a mixture containing all three compounds.
-
Can you elucidate the intricacies of cellular respiration, delineating the metabolic pathways involved and their respective roles in energy production within eukaryotic organisms ?
-
How do the mechanisms of ventilation and gas exchange operate synergistically in facilitating the diffusion of oxygen and carbon dioxide across the respiratory membrane, ensuring optimal...
-
How do environmental factors, such as altitude, temperature, and atmospheric composition, influence respiratory physiology, necessitating adaptive responses at both the cellular and systemic levels...

Study smarter with the SolutionInn App


