Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C
Question:
Give IUPAC names for the followingcompounds:
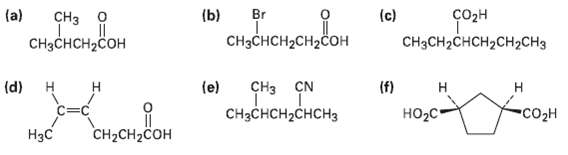
Transcribed Image Text:
(a) Br (b) еке (c) соон CHз о CH3CH2CHCH2CH2CH3 CHзснсH-CH2Coн CHзснCH2Cон (f) НO2с- (d) н CHз CN (e) н н Н CHзCнCH2CHсHз "Co2н CH2CH2COH Нзс
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Carboxylic acids are named by replacing e of the corresponding alkane with oic acid The carboxylic acid carbon is C1 WhenCOH is a substituent of a ring the suffix carboxylic acid is used the carboxyl ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the following compounds. (a) (b) (c) CH3 CH CH CH CH, CH, CH CH
-
A proposed project has fixed costs of $84,000 per year. The operating cash flow at 7,500 units is $93,000. Ignoring the effect of taxes, what is the degree of operating leverage? If units sold rise...
-
Is New Belgium Brewery centralized or decentralized? How does this make them more effective?
-
Match the following control frameworks with their main purposes. Purposes a. Expand internal controls to provide a broader view on risk management to maximize firm value. b. Provide management an...
-
Consider the two treatment assignment mechanisms (7.1) and (7.2). What is the probability \(P\left(T_{i}=1 ight)\) in each case? What is \(P\left(T_{i}=1 \mid Y_{i}(0)> ight.\) \(\left.Y_{i}(1)...
-
Nonprofit organizations routinely rely on generous corporate donations, and it is common to name facilities after benefactors. The Nationwide Childrens Hospital in Ohio is no exception; its name...
-
Constants The aurora is caused when electrons and protons, moving in the earth's magnetic field of 5.0 x 105T, collide with molecules of the atmosphere and cause them to glow. Part B What is the...
-
You work for ConstCo, a medium-sized engineering and construction company with annual profits of about $100 million. You recently entered as a contractor into a bidding process for a large...
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Draw structures corresponding to the following IUPAC names: (a) 2, 3-Dimethylhexanoic acid (b) 4-Methylpentanoic acid (c) Trans-1, 2-Cyclobutanedicarboxylic acid (d) o-Hydroxybenzoic acid (e) (9Z,...
-
Anthony is going to drive a flat-bed truck up a hill that makes an angle of 10 with respect to the horizontal direction. A 36.0 kg package sits in the back of the truck. The coefficient of static...
-
PLEASE USE CITATIONS APA FORMAT. include a document, news item, journal article or other reputable, verifiably accurate item related to the topic being discussed that unit week that has NOT already...
-
Answer the following questions https://www.youtube.com/watch?v=mZeNOaO3Pzk (Links to an external site.) Porter's Generic Business Unit Strategies Differentiation or Low Cost? Examine Amazon.com and...
-
What kind of user training should be conducted to deal with the issue of noise (electrical). How do you strike a balance between being overwhelmed with false positives and the danger of ignoring true...
-
Could you elucidate the concept of higher-order functions and delineate how they facilitate abstraction and enable functional programming paradigms?
-
Develop an incident and near miss reporting system for an organization. For your program, include the method for reporting of incidents and near misses, how staff will be trained on incident and near...
-
Consider the \(k^{\text {th }}\) moment functional given by \[T(F)=\int_{-\infty}^{\infty} t^{k} d F(t)\] where \(F \in \mathcal{F}\), a collection of distribution functions where \(\mu_{k}^{\prime}...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
How much heat (in kJ) is evolved in converting 1.00 mol of steam at 145 C to ice at -50 C? The heat capacity of steam is 2.01 J/g C, and that of ice is 2.09 J/g C.
-
Complete the following equations: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Phenol + NaH (k) Product of (j) + CH3OSO2OCH3 (l) Product of (j) + CH3I (m) Product of (j) + C6H5CH2Cl PhenolBr2 C, CS2...
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
A synthesis of the b-receptor blocker called toliprolol begins with a reaction between 3-methylphenol and epichlorohydrin. The synthesis is outlined below. Give the structures of the intermediates...
-
Use matrices (row-echelon form) to solve the following system of linear equa- tions. If the system has no solution, say that it is inconsistent. 3x + 2y = 7 x+y=3
-
For the following function : f(x) = 1/3 x^3 + 4x^2 + 16x a. Determine the critical points. b. Calculate the second derivative c. Determine if the function is concave up or down or not concave d....
-
2. An activity director for a cruise ship has surveyed 240 passengers. Of the 240 passengers; 135 like swimming, 150 like dancing, 65 like games, 80 like swimming and dancing, 40 like swimming and...

Study smarter with the SolutionInn App


