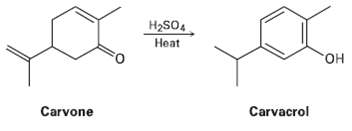
Heating carvone with aqueous sulfuric acid converts it into Carvacrol. Propose a mechanism for theisomerization. H2SO4 Heat
Question:
Heating carvone with aqueous sulfuric acid converts it into Carvacrol. Propose a mechanism for theisomerization.
Transcribed Image Text:
H2SO4 Heat "он Carvacrol Carvone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (21 reviews)
HA sa ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the rearrangement that converts an into an (Section 22.6). a-hydroxyimine a-aminoketone Nhemoglobin CH2NH hemoglobin AU: OK as changed? rearrangement CH2OH CH,OH
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for each of the following reactions: a. b. c. d. Br -, CH _CH3 0 H20 ( HOCH,CH,CH2 CHCH SCH2CH CH,CH-SH + --> CHCH2CCH3 N 2. HCI, H20
-
Inferring Investing and Financing Transactions and Preparing a Balance Sheet During its first week of operations ending January 7, 2011, FastTrack Sports Inc. completed six transactions with the...
-
In an age when millions of e-mails are sent daily, does the writing of an "old-fashioned" business letter potentially provide a competitive advantage or reflect favorably on the writer? Under what...
-
For which of the following ions does the formal charge give a fairly accurate picture of where the charge really is? Explain in each case. (a) NH4 (b) H3O: (C) NH2 (d) CH3
-
The soft-ride suspension system of the mountain bike is pinned at C and supported by the shock absorber BD. If it is designed to support a load of P = 1500 N, determine the factor of safety of pins B...
-
The December 31, 2015, balance sheet of the GAB LLP reads as follows: Capital is not a material income-producing factor for the LLP. Gina is an active (general) partner and owner of a 25% interest in...
-
Describe the process, advantages and applications of Resin Transfer Molding. What are the special features of Autoclave molding?
-
Randomized block design: Researchers interested in identifying the optimal planting density for a type of perennial grass performed the following randomized experiment: Ten different plots of land...
-
Amino acids can also be prepared by a two-step sequence that involves hell? Volhard?Zelinskii reaction of a carboxylic acid followed by treatment with ammonia. Show how you would prepare Leucine, (CH...
-
What is the irradiation at surfaces A 2 , A 3 , and A 4 of Example 12.1 due to emission from A 1 ?
-
Due to numerous complications involving missed medication dosages, you implement a study to determine the best strategy for enhancing medication adherence. Patients who are on a daily medication...
-
Johnny's Milkshakes is producing an income statement for last year. What category of expense should their utility costs (electricity) go under?
-
What is Photosynthesis?
-
An R-134a Refrigeration Syst m operates between 400 kPa (Evaporator) and 1.0 MPa (Condenser). The Internal Heat Exchanger (with HX = 90%) develops 50C. The compressor efficiency is 80%. Provide what...
-
Question 22 A m-4kg object at rest is taken from origin O(0,0) (m) to the point C(5,5) (m) by the path O-C line in a vertical plane from rest. During this movement, the forces acting on the object...
-
Assume that direct material costs in beginning Work-in-Process Inventory is $500 and an additional $1,500 worth of materials is added during the month. Equivalent units for direct materials is 500...
-
Let \(Y\) be \(\operatorname{binomial}(n=4=3)\). (a) Calculate the mean and variance by lling in the following table: i. \(\mathrm{E}[Y]=\) ii. \(\operatorname{Var}[Y]=\) (b) Calculate the mean and...
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at...
-
Spend a few minutes reviewing the Lewis dot symbols for the atoms H through Ne. Form a circle and have each group member ask the group member on his or her right to draw the Lewis symbol for a...
-
Show in detail curved-arrow the hydrolysis mechanism of N-methylbenzamide in aqueous NaOH. Assume that each mechanism involves a tetrahedral addition intermediate.
-
Give the structures of the hydrolysis products that result from each of the following reactions. (als)CH-1-0 110 NaOH
-
Use an analysis of resonance effects and leaving-group basicities to explain why acid-catalyzed hydrolysis of esters is faster than acid-catalyzed hydrolysis of amides.
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


