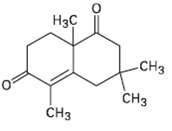
Question: How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and ?, ?-unsaturated ketone? Draw the structures of both
How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and ?, ?-unsaturated ketone? Draw the structures of both reactants and the intermediate Michael addition product.
.CH CH CH
Step by Step Solution
3.63 Rating (160 Votes )
There are 3 Steps involved in it
CH3CHCCHCH Michael acceptor H2O HC H Michael donor H3C CH3 CH3 C... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-C-R (22).docx
120 KBs Word File


