Identify each of the colored positions?red, blue, and green?as axial or equatorial. Then carry out a ring-flip,
Question:
Identify each of the colored positions?red, blue, and green?as axial or equatorial. Then carry out a ring-flip, and show the new positions occupied by each color,
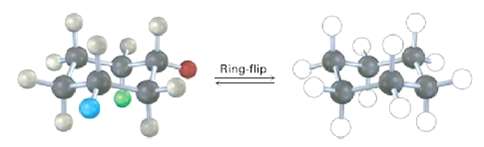
Transcribed Image Text:
Ring-flip
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
H I H I I H HB G equ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify each of the following as relating to (a) The control environment, (b) Control procedures, or (c) Monitoring. 1. Hiring of external auditors to review the adequacy of controls 2. Personnel...
-
Identify each of the following reactions as being a neutralization, precipitation, or oxidationreduction reaction. a. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g) b. Na2SO4(aq) + Hg(NO3)2(aq) HgSO4(s) +...
-
Identify each of the following elements as a metal, nonmetal, or metalloid: (a) Phosphorus (b) Strontium (c) Manganese, (d) Selenium (e) Sodium (f) Krypton.
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
How do a closed-end fund, an open-end fund, and an exchange-trade fund differ?
-
Apply the full White test for heteroskedasticity [see equation (8.19)] to equation (8.18). Using the chi-square form of the statistic, obtain the p-value. What do you conclude?
-
Using an example of static equilibrium of an element in pure two-dimensional (2D) shear stress, prove that the shear stresses are symmetric (i.e., prove that \(\sigma_{i j}=\sigma_{j i}\) when \(i eq...
-
A particular forecasting model was used to forecast a six- month period. Here are the forecasts and actual demands that resulted: Find the tracking signal and state whether you think the model being...
-
Please help describe one intervention that used a health behavior theory or model, that was effective in modifying the health behavior of high risk sexual behavior?
-
REPCO performs warranty repair work for name-brand kitchen appliances. REPCO bills appliance manufacturers on a costplus basis. It has a job-costing system that computes the cost of each order by...
-
Draw two different chair conformations of trans-1, 4-dimethylcyclohexane, and label all positions as axial or equatorial.
-
What is the energy difference between the axial and equatorial conformations of cyclohexanol (hydroxycyclohexane)?
-
How have the Internet, intranets, and extranets affected the types and uses of data resources available to business professionals? What other database trends are also affecting data resource...
-
1) Last week, your cafe sold 3,000 cups of coffee. Demand is expected to decrease by 10% for every 5% increase in price. Your current price is $2.80 and you are considering raising your price to...
-
Halley's comet has an orbital period of 74 earth years. Kepler's third law can be used to predict its orbital radius. What is its orbital radius in astronomical units? (one astronomical unit is...
-
A battery with e m f = 8 . 3 0 V and no internal resistance supplies current to the circuit shown in the figure below. When the double - throw switch S is open as shown in the figure, the current in...
-
A beer distributor sells 500 cases of regular 12-oz. Budweiser per week for 50 weeks each year - assume that there are 50 weeks in a year - from its warehouse in Central Austin. The distributor...
-
Consider a metal wire with a radius of 5 mm encased in plastic 1 mm thick. The plastic has a thermal conductivity of 0.2 W/m-K. If the wire must dissipate 10 W/m, what is the internal temperature...
-
P&G dubbed the internet research that consumers do prior to purchasing a product __________. a. FMOT b. ZMOT c. SMOT d. TMOT e. AMOT
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
A 50.0 g piece of iron at 152 C is dropped into 20.0 g H 2 O(l) at 89 C in an open, thermally insulatedcontainer. How much water would you expect to vaporize, assuming no water splashes out? The...
-
Starting with 2-methylpropene (isobutylene) and using any other needed reagents, outline a synthesis of each of the following (T = tritium, D = deuterium): (a) (b) (c) (d)
-
Show how you might carry out the following transformations: (a) (b) (c) (d) (e) OH Cl Ci OH
-
What compounds would you expect to be formed when each of the following ethers is refluxed with excess concentrated hydrobromic acid? (a) (b) (c) (d) (THF) (1,4-dioxane)
-
Convert the following ERDs to a Database schema. Follow all the steps in order as learned in this class. Identify PKs and FKs for each relation. Explain your work. Press esc to exit full screen Page...
-
How to draw a database in MS SQL Server for an entity with only a multivalued attribute and no primary key?
-
use Adventure Works Database Run each query and save the notebook with the results before submitting it. All the questions below are related to AdventureWorks database. make sure your notebook is...

Study smarter with the SolutionInn App


