Identify the following reactions as additions, eliminations, substitutions, orrearrangements: (a) CH3CH2Br + NACN CH3CH2CN (+ NaBr) (b)
Question:
Identify the following reactions as additions, eliminations, substitutions, orrearrangements:
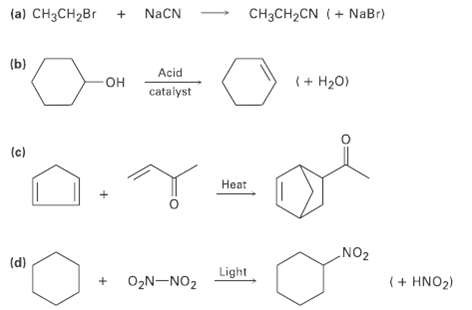
(a) CH3CH2Br + NACN CH3CH2CN (+ NaBr) (b) Acid (+ H20) OH cataiyst (c) Heat „NO2 "O. (d) Light (+ HNO2) O,N-NO2
Step by Step Answer:
a The reaction between bromoethane and sodium cyanide is a su...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Organic Chemistry questions
-
Identify each of the following reactions as fission, fusion, a transmutation, or radioactive decay. a. b. c. d. 4H He + 29e
-
Identify each of the following reactions as being a neutralization, precipitation, or oxidationreduction reaction. a. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g) b. Na2SO4(aq) + Hg(NO3)2(aq) HgSO4(s) +...
-
Sort the following reactions as either endothermic or exothermic. Endothermic Exothermic burning rocket fuel boiling water the reaction inside a chemical heat pack a burning match making popcorn in a...
-
If OPEC raised the price of oil high enough, would that be sufficient to promote an efficient energy mix?
-
Assume you have established an emergency fund and that you have saved an additional $12,000 that could fund an investment in common stock issued by the AT&T Corporation. Using the sources of...
-
The column is constructed from high-strength concrete and eight A992 steel reinforcing rods. If the column is subjected to an axial force of 200 kip, determine the average normal stress in the...
-
Consider the National Football League data in Table B.1. a. Use the forward selection algorithm to select a subset regression model. b. Use the backward elimination algorithm to select a subset...
-
James, who was engaged to examine the financial statements of Talbert Corporation, is about to audit payroll. Talbert uses a computer service center to process weekly payroll as follows. Each Monday...
-
M&R Company provided $ 2 , 0 0 0 in services to customers in December, which are not yet recorded. Those customers are expected to pay the company in January following the company s year - end. Wage...
-
Pink, Inc., is considering a project that will result in initial aftertax cash savings of $4.1 million at the end of the first year, and these savings will grow at a rate of 3 percent per year...
-
Identify the functional groups in the following molecules, and show the polarity ofeach: (a) CH3CH2C=N (c) () OCH3 CCH-c (f) (e) (d) "NH2
-
What is the difference between a transition state and an intermediate?
-
If f is continuous and 31 f(x) dx = 8, show that f takes on the value 4 at least once on the interval [1, 3].
-
An investor buys 200,000 shares of a private placement that's sold under Regulation D. The investor is not an officer or director of the company. How many shares is the investor permitted to sell...
-
What are some key performance indicators (KPIs) used in the accounting and financial analysis of a manufacturing company?
-
Consider how well jewelry sales can be forecast on the basis of disposable personal income or unemployment rate, as measures of overall purchasing power. The explanatory variables selected are based...
-
Match the words with the term Question 1 options: semi-variable costs difference between revenue and total costs variable costs non-cash expense How are manufacturing overhead costs allocated and...
-
As of January 2020, the average pay for a Level 1 Registered Nurse (RN) is $63,190 annually. Leo is a registered nurse (RN) with gross earnings of $2,334.04 semimonthly. If the hospital where Leo...
-
A certain Mr. Claus has mixed up his Scandinavian gift bags this Christmas, and he does not know which one goes to Denmark, which one goes to Sweden, and which one goes to Norway. The Danish bag has...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
The bond angle in H 2 Se is best described as (a) Between 109 and 120; (b) Less than in H 2 S; (c) Less than in H 2 S, but not less than 90; (d) Less than 90.
-
Starting with styrene, outline a synthesis of each of the following: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) Cl Cl CHs OH OH C&Hs OH OH C6H5 Br C6H5 caHs CeHs C6H5
-
Starting with toluene, outline a synthesis of each of the following: (a) m-Chlorobenzoic acid (b) p-Methylacetophenone (c) 2-Bromo-4-nitrotoluene (d) p-Bromobenzoic acid (e)...
-
Starting with aniline, outline a synthesis of each of the following: (a) p-Bromoaniline (b) o-Bromoaniline (c) 2-Bromo-4-nitroaniline (d) 4-Bromo-2-nitroaniline (e) 2,4,6-Tribromoaniline
-
123 Anna purchased 100 shares of Delta stock on February 1, Year 2, for $46 per share, and 5 received a two-for-one stock split on December 31, Year 2. Anna sold all the shares of Delta stock on...
-
work i Saved QS 17-10 (Algo) Computing activity rates for activity-based costing LO P3 A company sells two types of products: standard and deluxe. It prepares the following analysis showing budgeted...
-
! Required information [The following information applies to the questions displayed below.] Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It...

Study smarter with the SolutionInn App


