Identify the functional groups in each of the following molecules: (a) Methionine, an amino acid: (b) Ibuprofen,
Question:
Identify the functional groups in each of the following molecules:
(a) Methionine, an amino acid:
(b) Ibuprofen, a pain reliever:
(c) Capsaicin, the pungent substance in chilipeppers:
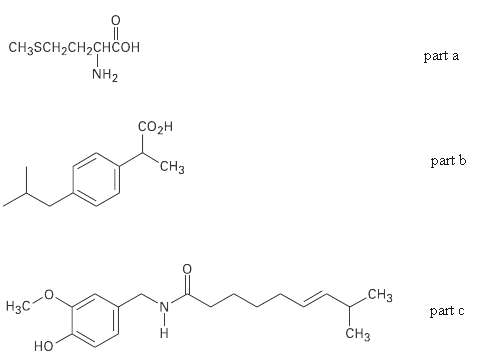
Transcribed Image Text:
|| CHgSCH-CH2CHCон part a NH2 созн part b CH3 CHз Нзс N. part c CHз Н но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Notice that certain functional groups have different designations i...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the functional groups in each of the following molecules: (a) CH3CH2COCH2CH2CH3 (b) CH3COOC2H5 (c) CH3CH2OCH2CH2CH2CH3
-
Identify the functional groups in each of the following compounds: CH2CH2CH2CH2CH2CH3 Cl OH CH2CH2CH CH H2C CH3CH2CH2CH2 CH2CH2CH2CH3
-
Identify all of the functional groups in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) Vitamin D3 HO OMe Aspartame O NH2 NH2 Amphetamine Me Cholesterol HO OCH2CH3 Demerol CH A...
-
Design a linear-time algorithm to sort an array of Comparable objects that is known to have at most three distinct values. (Edsger Dijkstra named this the Dutch-national-flag problem because the...
-
Do you agree that a mission statement provides substantive guidance while a vision statement provides inspirational guidance? Explain.
-
Find i, in the circuit of Fig. 4.9 using superposition. 292 ww 4 A 392 592 wwww www Figure 4.9 For Example 4.4. 192 www 20 V 5i www 492
-
The mean room and board expense per year at four-year colleges is \($10,453\). You randomly select 9 four-year colleges. What is the probability that the mean room and board is less than \($10,750?\)...
-
The Tiny Treasures Company must decide whether to continue selling a line of childrens shoes manufactured on a machine that has no other purpose. The machine has a current book value of $12,000, and...
-
Bond X is a premium $1000 par value bond making annual payments. The bond has a coupon rate of 9%, a YTM of 7%, and has 13 years to maturity. Bond Y is a discount $1000 par value bond making annual...
-
From the first quarter 2018 I 0-Q of Groupon, Inc.: Groupon operates online local commerce marketplaces throughout the world that connect merchants to consumers by offering goods and services,...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Propose structures for simple molecules that contain the following functional groups: (a) Alcohol (b) Aromatic ring (c) Carboxylic acid (d) Amine (e) Both ketone and amine (f) Two double bonds
-
The status of a pension plan as reported in its financial statements typically differs significantly from its status as presented in the required supplementary information for the plan. Why? Do you...
-
Elaborate on the branches of civil engineering 1. Water Resources Engineering 2. Transportation Engineering 3. Environmental Engineering
-
Carri and Dane, ages 34 and 32, respectively, have been married for 11 years, and both are active participants in employer-qualified retirement plans. Their total AGI in 2023 is $231,000, and they...
-
Using figures from the trial balance, compute total assets, total liabilities, and owners' equity. Total assets Total liabilities Total owners' (stockholders') equity S 248,500 I < Transaction...
-
Demonstrates a thorough understanding of Consignment Sales Accounting. required 5 current sources, of which at least 2 are from journal articles. All web sites utilized are authoritative. Cites all...
-
Which would best help the external auditor evaluate the objectivity of the client's internal auditor? Group of answer choices Evaluate the quality of the internal auditor's documentation Determine if...
-
Develop the appropriate primary research question to be associated with this design. Develop a hypothetical research scenario that would necessitate the use of a Longitudinal Design. The research...
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
Assume that a skater has a mass of 80 kg and that his skates make contact with 2.5 cm 2 of ice. (a) Calculate the pressure in atm exerted by the skates on the ice. (b) If the melting point of ice...
-
Write structural formulas for all the products that would be obtained when each of the following alkyl halides is heated with potassium tert-butoxide in tert-butyl alcohol. When more than one product...
-
Starting with an appropriate alkyl halide and base, outline syntheses that would yield each of the following alkenes as the major (or only) product: (a) (b) (c) (d) (e)
-
Predict the more stable alkene of each pair: (a) 2-methyl-2-pentene or 2,3-dimethyl- 2-butene; (b) Cis-3-hexene or trans-3-hexene; (c) 1-hexene or cis-3-hexene; (d) Trans-2- hexene or...
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


