In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as
Question:
In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as shown. Analyze the stereochemistry of the reaction in this case and explain which products areformed.
Transcribed Image Text:
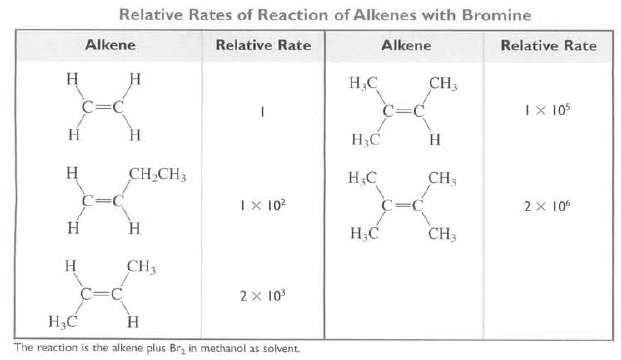
Relative Rates of Reaction of Alkenes with Bromine Alkene Relative Rate Alkene Relative Rate Н CH3 Н.С C=C IX 105 Н Н Н С Н Н CH CH3 CH3 НС C=C IX 10? 2 x 10 Н Н Н.С CH3 CH3 Н 2 x 10 H3C н The reaction is the alkene plus Br, in mathanol as solvent.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
If Br 2 adds from the bottom rather than the as shown in Figur...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The rate law for addition of Br2 to an alkene is first order in Br2 and first order in the alkene. Does this fact prove that the mechanism of addition of Br2 to an alkene proceeds in the same manner...
-
The gas-phase reaction between Br2 and H2 to form HBr is assumed to proceed by the following mechanism: a. Under what conditions does the rate law have the form rate = k[Br2]? b. Under what...
-
From what you know about the stereochemistry of alkene addition reactions, predict the configurations of the products that would be obtained from the reaction of 2-butyne with the following: a. One...
-
0 3 S 25 cos(x) sin (x) dx -T3
-
Graywall, CPA, serves as a trustee of the CureCancerNow Foundation, a charitable foundation that is dedicated to cancer research activities. For years, he has been the loyal friend and advisor to...
-
When a nerve impulse propagates along a nerve cell, the electric field within the cell membrane changes from 7.0 105 N/C in one direction to 3.0 105 N/C in the other direction. Approximating the...
-
Suppose that, as in the corn farm example, the farm has random production and the final spot price is governed by the same demand function. However, the crop of the farm is not perfectly correlated...
-
Bernadette, a longtime client of yours, is an architect and the president of the local Rotary chapter. To keep up to date with the latest developments in her profession, she attends continuing...
-
Sultan Company uses an activity-based costing system. At the beginning of the year, the company made the following estimates of cost and activity for its five activity cost pools: Activity Cost Pool...
-
Information about Zhu Boards is presented in E6-4. Additional data regarding Zhu's sales of Xpert snowboards are provided below. Assume that Zhu uses a perpetual inventory system. Instructions (a)...
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
Explain which of the three products shown in is formed when 1-butene reacts with HCI.
-
Crazy Freddies is a mail order and Internet-based electronic retailer incorporated and based in Grand Rapids, Michigan. Crazy Freddies takes significant measures to avoid triggering income tax nexus...
-
Why must some of the equipment and people be idle some of the time for the plant to be effective? Explain
-
Which of the following are modern approaches of the management of international services and manufacturing operations? business process redesign (BPR), six sigma, lean manufacturing, and...
-
Discuss the difference between the policies and procedures. Identify and discuss the different types of Standard Operating Procedures and Standard Operating Guidelines, relative to enhanced...
-
__________ companies often centralize staff functions to support all business units. ___________ companies often decentralize staff functions, dedicating them to each business or product...
-
Cristol Company produces sheets. The master budget shows the following standards information and indicates the company expected to produce and sell 27,000 units for the year. Direct Materials 3 yards...
-
In a letter to a member of Congress, Fed Chairman Ben Bernanke made the following statement: The monetary accommodation provided by the Federal Reserve has substantially helped the U.S. economy by...
-
Describe basic managerial approaches to implementing controls and how these are implemented.
-
A first-order reaction has rate constants of 4.6 10 -2 s -1 and 8.1 10 -2 s -1 at 0C and 20.C, respectively. What is the value of the activation energy?
-
To what classes do the following enzymes belong? (a) Pyruvate decarboxylase (b) Chymotrypsin (c) Alcohol dehydrogenase
-
Identify the following aminoacids: (a) (b) (c)
-
Give the sequence of the following tetra peptide (yellow =S):
-
The government standard on radiation from electrical devices is 10mW/cm2 (maximum). Assume a laptop computer is found to radiate more than this standard. The electric field inside is measured to be 5...
-
A 2 2 . 8 - m deep pool is filled with oil of density 8 9 5 kg / m ^ 3 . What is the gauge pressure at the bottom of the pool? Part B: What is the force from the okl that exerts on a 0 . 3 5 m x 0 ....
-
Clark was driving his four-wheeler 15 meters per second on a dirt road. It had just rained and there were massive mud puddles on the road. He hits on and it slows him to a speed of 3.7 meters per...

Study smarter with the SolutionInn App


