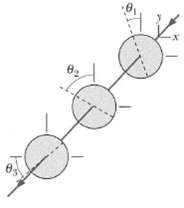
Question: In Figure, unpolarized light is sent into a system of three polarizing sheets with polarizing directions at angles 1 = 20?, 2 = 60?, and
In Figure, unpolarized light is sent into a system of three polarizing sheets with polarizing directions at angles θ1 = 20?, θ2 = 60?, and θ3 = 40?. What fraction of the initial light intensity emerges from the system?
Step by Step Solution
3.35 Rating (155 Votes )
There are 3 Steps involved in it
We apply Eq 3340 once and ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
2-P-E-E-W (133).docx
120 KBs Word File


