Isoborneol (Problem 27.37) is converted into camphene on treatment with dilute sulfuric acid. Propose a mechanism for
Question:
Isoborneol (Problem 27.37) is converted into camphene on treatment with dilute sulfuric acid. Propose a mechanism for the reaction, which involves a carbocationrearrangement.
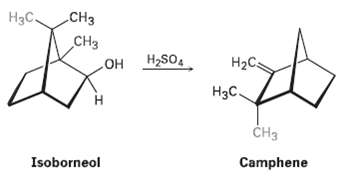
Transcribed Image Text:
Cнз Нас. сHз H2SO4 HO НаС Нас. ČH3 Camphene Isoborneol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
OH X2XX H loss of H30 Isoborneol HC OH H3C HOH ...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Propose a mechanism for the following reaction: + HC-o trace H CH5
-
6. Kindly describe the accounting framework. What are the elements and how do they effect the financial statements? 7. What does the accounting model specify? What are the elements involved?
-
What is a strategic network? What is a strategic center firm? How is a strategic center used in business-level, corporate-level, and international cooperative strategies? Discuss.
-
Referring to the figure, assume the flow to be friction less in the siphon. Find the rate of discharge in cubic feet per second, and the pressure head at B if the pipe has a uniform diameter of 1 in....
-
McDonald and Ayers [1978] present data from an early study that examined the possible link between air pollution and mortality. Table B. 15 summarizes the data. The response MORT is the total...
-
Hill Street Electronics has provided the following information for last year: Sales revenue . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 8,000,000 Store rent . . . . . . ....
-
Sheridan Corp. had total variable costs of $224.200 total hied costs of $143500, and total revenues of $380.000. Compute the required sales in dollars to break even
-
Prepare a simple tax provision and the journal entries to calculate the current and the deferred tax provision (benefit) using the following data: a. ABC, Inc. reported pre-tax book income of...
-
Propose a mechanistic pathway for the biosynthesis of isoborneol. A carbocation rearrangement is needed at one point in thescheme. CH CH HO Isoborneol
-
Digit oxigenin is a heart stimulant obtained from the purple foxglove Digitalis purpurea and used in the treatment of heart disease. Draw the three-dimensional conformation of digitoxigenin, and...
-
Interest rates on 4-year Treasury securities are currently 7%, while 6-year Treasury securities yield 7.5%. If the pure expectations theory is correct, what does the market believe that 2-year...
-
Is this done correctly? Assume an investor purchases the business of an investee for $26,880. The fair values of the individual net assets are equal to their reported book values. The investee...
-
remember that nuveen agreed to purchase a $250 million (face amount) bond. the current value of the bond is $234,800,010 and the price paid upon issuance was $207,827,221. what is the paper gain...
-
ABC rental posted the following monthly rent per type of accommodation: Rent for month One-bedroom apartment $1250 Two-bedroom apartment $1650 Three-bedroom apartment $1900 If the company has 70...
-
Valley Hospital's bed count was 150 from June 1st through June 15th. On June 15th the bed count changed to 175. The inpatient service days for the month are 3,978. What is the percentage of occupancy...
-
An airplane maintains a heading due east at an air speed of 750 km/h. It is flying through heavy winds of 200.0 km/h, from the south. (a) What is the plane's ground speed? (b) In which direction is...
-
How do we calculate the firms free cash flow, and the free cash flow to equity?
-
Classify each of the following as direct costs or indirect costs of operating the Pediatrics ward for children at the Cleveland Clinic: a. Wi-Fi covering the entire hospital campus b. Net cost of...
-
Geologists can estimate the age of rocks by their uranium-238 content. The uranium is incorporated in the rock as it hardens and then decays with first-order kinetics and a half-life of 4.5 billion...
-
Write an equation for the reaction of d-mannose with Fehling's reagent (Cu2+) to give d-mannonic acid.
-
Write the structure of d-mannaric acid.
-
Write an equation for the acid-catalyzed reaction of b-d-galactose with methanol.
-
1. Mention five key organizational characteristics that the Cll research team identified for organizations with effective quality management systems. provide brief discussion of each and how it...
-
When a metal was exposed to photons at a frequency of 1.46 1015 s1, electrons were emitted with a maximum kinetic energy 3.60 10-19 J. G ? Calculate the work function, , of this metal. J/photon What...
-
No. 101 Debit Cash $ 2,400 Question 2 (10 pts) On November 1, 2024, the account balances of Schilling Equipment Repair were as follows. Accumulated Depreciation-Equipment A Clipboard Font Alignment...

Study smarter with the SolutionInn App


