Lactate, a product of glucose catabolism in oxygen-starved muscles, can be converted into pyruvate by oxidation. What
Question:
Lactate, a product of glucose catabolism in oxygen-starved muscles, can be converted into pyruvate by oxidation. What coenzyme do you think is needed? Write the equation in the normal biochemical format using a curvedarrow.
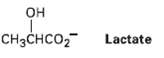
Transcribed Image Text:
OH Lactate CHзснCO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
OH CH3CHCO Lactate NAD NADHH ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What market research techniques do you think is most effective and least effective? Are there conditions under which some market research techniques work better than others? If so, what are those...
-
What type of business strategy do you think is most compatible with a high-performance work system? Why?
-
What social problem do you think is the worlds biggest? Wars? Global warming? Resource imbalances? How could you start to solve a big social problem through marketing?
-
5.In a nut shell, when you look back over time, the power of image has always been there. Even in the prehistoric era, they used imagery to communicate, and what's even more incredible is that we are...
-
Note the enterprise and equity valuations for Life Technology in the Excel spreadsheet model entitled Target Valuation Model on the companion website accompanying this book. View this as the base...
-
A starship is circling a distant planet of radius R. The astronauts find that the free-fall acceleration at their altitude is half the value at the planets surface. How far above the surface are they...
-
The following data were collected on specific gravity and spectrophotometer analysis for 26 mixtures of NG (nitroglycerine), TA (triacetin), and 2 NDPA (2-nitrodiphenylamine). There is a need to...
-
The following data represent the responses to two questions asked in a survey of 40 college students majoring in business: What is your gender? (M = male; F = female) and What is your major? (A =...
-
A set S R^n is convex if for all x, y S and t [0, 1], we have (1 t)x + t y S (in other words, if S contains the points x and y, it contains the entire line segment between them). (a) If W R^n is a...
-
The following salaried employees of Mountain Stone Brewery in Fort Collins, Colorado, are paid semimonthly. Some employees have union dues or garnishments deducted from their pay. Required: Calculate...
-
Why arent the glycolysis and gluconeogenesis pathways the exact reverse of one another?
-
How many moles of acetyl CoA are produced by catabolism of the following substances? (a) 1.0 mol glucose (b) 1.0 mol palmitic acid (c) 1.0 mol maltose
-
Find the circumference and area of the circle. Round to the nearest tenth. 12 in.
-
The rule of seven is based on the use of continuous interest to approximate the doubling time of money. The "7" comes from the fact that the natural log of 2 is approximately 0.7. The rule says that...
-
1. Calculate the mass of a molecule of N2O3. The atomic masses of N and O are 14.01 amu/atom and 16.00 amu/atom respectively. 2. Calculate the mass of 4.8 mol of N2(H5C3)4. The molar masses of N, H...
-
Oxford healthcare receives $3,000 in checks daily which are cleared in 3 days and writes $2,000 in checks daily and this takes 6 days for these to be received and clear the banking system. What is...
-
If an insitution says that the interest rate is r% compounded monthly, we say that r% is the nominal rate of interest. The effective rate is the rate that earns the equivalent amount at the end of...
-
3. A new startup wants to test drive their amazing new product (edible laundry pods!) next summer. The have a forecast for the demand (d1, d2, d3) and the production costs (c1, c2, c3) for each of...
-
Use the reversal property to show that \[ \mathscr{F}\left\{A e^{-\alpha t \mid} \operatorname{sgn}(t)ight\}=\frac{-2 A j \omega}{\omega^{2}+\alpha^{2}} \]
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
One of the purposes of the kidneys is to transfer useful chemicals from the urine to the blood, and toxins from the blood to the urine. In the transport of glucose from the urine to the blood, the...
-
Acid-catalyzed dehydration of 2,2-dimethyl-1-hexanol gave a number of isomeric alkenes including 2-methyl-2-heptene as shown in the following formula. (a) Write a stepwise mechanism for the formation...
-
Compound A (C4H10) gives two different monochlorides on photochemical chlorination. Treatment of either of these monochlorides with potassium tert-butoxide in dimethyl sulfoxide gives the same alkene...
-
Compound A (C6H14) gives three different monochlorides on photochemical chlorination. One of these monochlorides is inert to E2 elimination. The other two monochlorides yield the same alkene B...
-
What measures can organizations implement to promote transparency and accountability in the budgeting process, thereby fostering trust and buy-in among stakeholders and facilitating...
-
Based on the following cost expenditures for the construction of an office / warehouse development ( to include direct and indirect charges ) , calculate the peak financial requirement: Month Monthly...
-
Carlos has worked very hard through his career as a teacher and is contemplating an early retirement. He already has a pension in place commencing at retirement age but wants to cut back on his...

Study smarter with the SolutionInn App


