Question: List the point coordinates for all atoms that are associated with the FCC unit cell(figure). (b) (a) (c)
List the point coordinates for all atoms that are associated with the FCC unit cell(figure).
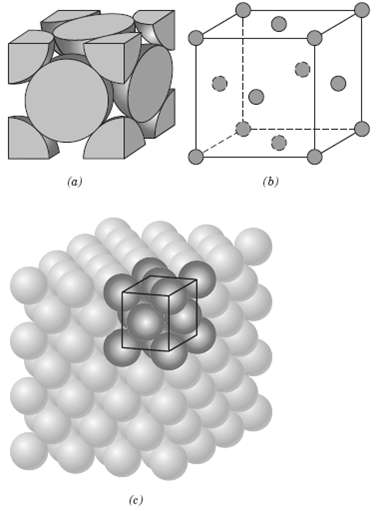
(b) (a) (c)
Step by Step Solution
3.28 Rating (169 Votes )
There are 3 Steps involved in it
From figure b the atom located of the origin of the unit cell h... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
33-E-M-S-E-M-S (45).docx
120 KBs Word File


