Look at figure, and tell which molecular orbital is the HOMO and which is the LUMO for
Question:
Look at figure, and tell which molecular orbital is the HOMO and which is the LUMO for both ground and excited states of ethylene and 1, 3-butadiene.
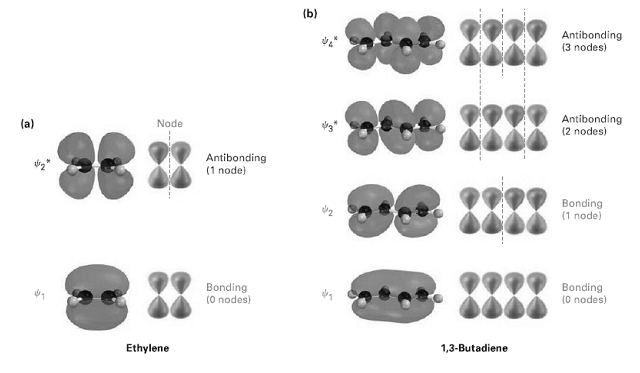
Transcribed Image Text:
(b) Antibonding (3 nodes) Antibonding (2 nodes) (a) Node Antibonding (1 node) Bonding (1 node) Bonding (0 nodes) Bonding (0 nodes) 1,3-Butadiene Ethylene ------- -- -----
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
For ethylene Energy 88 Two 2p atomic orbitals 42 LUMO Antib...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which molecular orbital of ethylene ( or *) is the most important one to look at in a reaction in which ethylene is attacked by an electrophile?
-
Look at the following orbital diagrams and electron configurations. Which are possible and which are not, according to the Pauli exclusion principle? Explain. a. b. c. d. 1s22s22p4 e. 1s22s42p2 f....
-
Ethylene (C2H4) at 25oC and 1 atm is burned with 300% excess air at 25oC and 1 atm. Assuming that this reaction takes place adiabatically at 25oC and that the products leave at 25oC and 1 atm....
-
On January 2, $217981 in 10-year, 5% bonds with a market interest rate of 9%, and interest payable semiannually, were issued for $185895. On June 30, bond interest was paid. On December 31, the...
-
Why did Citibank and Travelers resort to a co-CEO arrangement when they merged in 1998? What are the advantages and disadvantages of such an arrangement?
-
Exhibit 3.32 presents a statement of cash flows for Walmart for fiscal 2015, 2014, and 2013. This statement matches the Walmart statement of cash flows in Appendix A, and is an expanded version of...
-
The probability that the noise level of a wide-band amplifier will exceed \(2 \mathrm{~dB}\) is 0.05. Use Table 1 or software to find the probabilities that among 12 such amplifiers the noise level...
-
The Ajax Coal Company is the only employer in its area. It can hire any number of female workers or male workers it wishes. The supply curve for women is given by Lf = 100wf MEf = Lf =50 and for men...
-
A lake with a fixed carrying capacity contains a certain fish population. The fish population in the lake has a growth rate that is proportional to its size when the population is very small relative...
-
How might one employee coach or train another employee by use of Twitter and text messaging?
-
The amino acid cysteine, C3H7NO2S, is biosynthesized from a substance called cystathionine by a multistep pathway. (a) The first step is a transamination. What is the product? (b) The second step is...
-
Draw the products you would expect from conrotatory and disrotatory Cyclizations of (2Z, 4Z, 6Z)-2, 4, 6-octatrienc. Which of the Iwo paths would you expect the thermal reaction to follow?
-
Again consider the stock market poll discussed in Exercise 7.47. a. Suppose we wish to use the poll's results to justify the claim that fewer than 50 percent of American investors in 1992 found the...
-
An inchworm can travel a distance of 3 yards in its lifetime. What is this distance in cm? Recall (1 yard = 3ft) and (1 inch = 2.54cm)
-
A motor turns its shaft counterclockwise with a torque of 5Nm. this turns a wheel with a diameter of 1m attached to the shaft. This pulls up a chain which is attached to a 40cm lever. The lever has a...
-
A simple frictionless pulley hanging from the ceiling of my garage and lifting a mass of 10kg. The pulley has a diameter of 3m, the rope has a diameter of 0.01m. I pulled down on the right side of...
-
A student leaving campus for spring break wants to make sure the air in his dorm room has a high water vapor pressure so that his plants are comfortable. The dorm room measures 3.45 m 4.64 m 3.63 m...
-
The internal forces within the train (the forces each car exerts on its neighbors) are equal and opposite, according to Newton's third law. This means they cancel each other out when considering the...
-
Redo question 42 using the data for Ford. Question 42 Estimate the regression of Chryslers return on assets against its debt/equity ratio. Compute the DurbinWatson statistic. Does autocorrelation...
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
High frequency trading is the practice of executing multiple transactions for securities followed by extremely short holding periods. Such market practices often require highly sophisticated...
-
Which of the isomeric alcohols having the molecular formula C5H12O are chiral? Which are achiral?
-
Write structural formulas or make molecular models for all the compounds that are trichloro derivatives of cyclopropane. (Don't forget to include stereoisomers.) Which are chiral? Which are achiral?
-
In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. a. b. c. d. CICH CHCH OH and HOCH CHCH,OH CI OH CH,CH...
-
The community nursing service team provides nursing care to patients in the community, Discuss?
-
Why are angiosperms so diverse (when compared with non-flowering lineages of plants)? How expensive flowers are to produce for the plant (they cost lots of ATP to make); what benefit do they provide...
-
Martinez Company's relevant range of production is 7,500 units to 12,500 units. When it produces and sells 10,000 units, its average costs per unit are as follows: Average Cost Per Unit Direct labor...

Study smarter with the SolutionInn App


