Magnesium oxide has the rock salt crystal structure and a density of 3.58 g/cm 3 . (a)
Question:
Magnesium oxide has the rock salt crystal structure and a density of 3.58 g/cm3.
(a) Determine the unit cell edge length.
(b) How does this result compare with the edge length as determined from the radii in Table 12.3, assuming that the Mg2+ and O2- ions just touch each other along the edges?
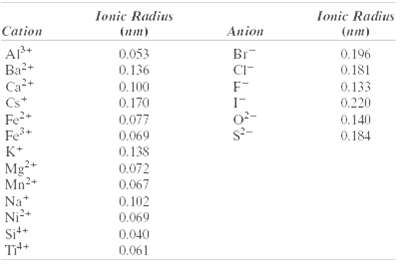
Transcribed Image Text:
Ionic Radius fonic Radius Cation Anion (nm) (nm) AP+ Ba+ Ca+ Br 0.053 0.196 0.181 0.136 CI- 0.133 0,220 F- 0.100 0.170 Cs* Fe+ Fe K+ 0.077 0.140 0.184 0.069 0.138 0.072 0.067 Mg+ Mn2+ Na* Ni² Sit+ 0.102 0.069 0.040 0.061
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
a This part of the problem calls for us to determine the ...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Iron oxide (FeO) has the rock salt crystal structure and a density of 5.70 g/cm3. (a) Determine the unit cell edge length. (b) How does this result compare with the edge length as determined from the...
-
Magnesium (Mg) has an HCP crystal structure and a density of 1.74 g/cm3. (a) What is the volume of its unit cell in cubic centimeters? (b) If the c/a ratio is 1.624, compute the values of c and a.
-
Zirconium has an HCP crystal structure and a density of 6.51 g/cm 3 . (a) What is the volume of its unit cell in cubic meters? (b) If the c/a ratio is 1.593, compute the values of c and a.
-
True or False? Azure files can be accessed from anywhere in the world using a URL that points to the file. True False
-
Overall, studies have shown that stereotypes most often are accurate, with some notable exceptions. For example, political stereotypes are consistently inaccurate, while gender stereotypes tend to be...
-
The balance sheets for Federer Sports Apparel for 2022 and 2021 are presented below. Required: 1. Prepare a vertical analysis of the balance sheet data for 2022 and 2021. Express each amount as a...
-
Find the solution to the Laplace equation in \(R^{3}\) : \[\frac{\partial^{2} u}{\partial x^{2}}+\frac{\partial^{2} u}{\partial y^{2}}+\frac{\partial^{2} u}{\partial z^{2}}=0\] assuming that \[u(x,...
-
At the end of June, Morton Company had a balance of $49,900 in the vacation benefits payable account. During July, employees earned an additional $3,110 in vacation benefits, but some employees used...
-
2. A Corporation issues a bond with a face value of $250,000 and a stated rate of 8% interest. The interest is paid annually during the bond's 10-year life. The market rate for a similar bond is 6%....
-
Map the EER diagrams in Figures 4.9 and 4.12 into relational schemas. Justify your choice of mapping options. Figures 4.9 Figure 4.12 Salary Shift Model Capacity Weight M. WORKS ON N EMPLOYEE N...
-
Calculate the density of FeO, given that it has the rock salt crystal structure.
-
Compute the theoretical density of diamond given that the CC distance and bond angle are 0.154 nm and 109.5, respectively. How does this value compare with the measured density?
-
When firms allocate service department costs to production departments, why would they first accumulate these costs at the service department level rather than assigning them directly to production...
-
A good way to make sure you understand organizational politics is to practice enacting different political tactics, and to attempt to recognize these tactics portrayed by others. This exercise is...
-
What is empowerment? Can it work on the team as well as individual level?
-
After General Manuel Noriega had come under the authority of the United States forces following the invasion of Panama, his attorneys argued that he should be treated as a prisoner of war (POW) under...
-
What is the timeline for the U.S. federal budget each year? When does a fiscal year begin and end?
-
Any training course on the essentials of customer service will advise you always to smile at customers and to make eye contact with them. In fact, it seems so commonsensical as to not need repeating....
-
What career opportunities in sales are offered by service companies?
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
What property of diamond leads to the most engineering applications? Which types of applications would benefit from this property?
-
Is a brittle material a weak material?
-
What is the toughness of a material?
-
What is the difference between true stress and engineering stress? True strain and engineering strain?
-
How does the 360-degree feedback method address the court's six stress issues depicted in Gerhart and Newman's reading? Since the Brito case, there has been growing evidence that the courts have very...
-
Read"Why Race Isn't as"Black"and White"as We Think"by Brent Staples. What can you find out about your own racial,religious,and cultural heritage? Search the Gateway library database: Ancestry Library...
-
Describe the classic steps for identifying an endocrine gland. Are these steps practical for identifying the sources of all the hormones we know of today? Explain.

Study smarter with the SolutionInn App


