Make a copy of the isothermal transformation diagram for a 0.45 wt% C iron-carbon alloy (Figure), and
Question:
Make a copy of the isothermal transformation diagram for a 0.45 wt% C iron-carbon alloy (Figure), and then sketch and label on this diagram the time-temperature paths to produce the following microstructures:
(a) 42% proeutectoid ferrite and 58% coarse pearlite
(b) 50% fine pearlite and 50% bainite
(c)100% martensite
(d) 50% martensite and 50% austenite
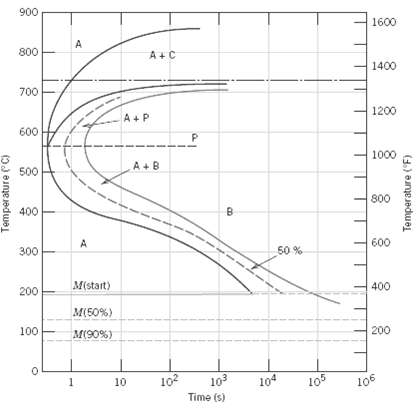
Transcribed Image Text:
900 1600 800 1400 700 1200 A + P 600 1000 A +B 500 800 400 600 50 % 300 M(start) 400 200- M(50%) 200 100 M(90%) 10? 103 104 10 106 10 Time (s) Temperature (C) Temperature (F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Below is shown an isothermal transformation diagram for a 045 wt C ironcarbon alloy with t...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Make a copy of the isothermal transformation diagram for an iron-carbon alloy of eutectoid composition (Figure 10.22) and then sketch and label time-temperature paths on this diagram to produce the...
-
Make a copy of the isothermal transformation diagram for an iron???carbon alloy of eutectoid composition (Figure) and then sketch and label time???temperature paths on this diagram to produce the...
-
Make a copy of the following accounts. Balance each account as appropriate. Dr Motor vehicles Cr 7 000 Aug 9 500 Aug Balance 12 Bank 7 000 14 Rank Dr Bank loan Cr Aug 31 Bank 3 200 Aug 1 Balance 39...
-
Data for Barry Computer Co. and its industry averages follow. a. Calculate the indicated ratios for Barry. b. Construct the DuPont equation for both Barry and the industry. c. Outline Barrys...
-
Is the selection process for determining members of the National Labor Relations Board too politicized? Explain your response. Does the current selection process lead to instability in...
-
Prepare journal entries to record each of the transactions listed in E 21. Data From Problem E 2-1 1. Issued 30,000 shares of common stock in exchange for $300,000 in cash. 2. Purchased equipment at...
-
What does it mean to recommend that an expense or revenue should be "accrued?" Would this be considered "normal?"
-
Circular copper rod of diameter D = 1 mm and length L = 25 mm are used to enhance heat transfer from a surface that is maintained at Ts.1 = 100C. One end of the rod is attached to this surface (at x...
-
Suppose that a bushel of wool could be traded for a barrel of wine, one-for-one, on the international market. Draw a diagram illustrating the original situation, and this new situation, with wool on...
-
Ziggy's Inc. is a retailer of highly specialized clothing that opened for business on May 1, 2014, in a leased store in a strip mall. A condensed income statement for Ziggy's Inc. for its year, ended...
-
For parts (a), (c), (d), (f), and (h) of Problem 10.20, determine the approximate percentages of the microconstituents that form. (a) Rapidly cool to 250(C (480(F), hold for 103 s, then quench to...
-
Name the microstructural products of eutectoid ironcarbon alloy (0.76 wt% C) specimens that are first completely transformed to austenite, then cooled to room temperature at the following rates: (a)...
-
The number N of locations of a popular coffeehouse chain is given in the table. (The numbers of locations as of June 30 are given.) (a) Find the average rate of growth (i) From 2000 to 2002 (ii) From...
-
Classify each of the following items as excludable, nonexcludable, rival, or nonrival. Oxygen A bottle of wine A view of a harbor A public square A Facebook post
-
A friend of yours who has been pursuing pottery as a hobby has just formed a business selling a range of gift items. Advise the founder on her options for distributing the companys products.
-
Henderson worked as a chemical engineer for the Wannisky Chemical Corporation. McGuire, Hendersons supervisor, ordered him to remove the labels from several hundred steel drums that had once...
-
The following lead paragraph was published in an article entitled, Why the Times Could Go Private, in the December 11, 2006 issue of Business- Week : Even before restive shareholders began ramping up...
-
Through Langham Engineering, Michael Langham marketed his invention, the cross-slope monitor (CSM), as an accessory to John Deere equipment. He had a plan to expand his business to include the CSM as...
-
Mary Rossi, a student at State College, has a balance of $380 on her retail charge card; if the store levies a finance charge of 21% per year, how much monthly interest will be added to her account?
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Cylinders of compressed gas are equipped with pressure gauges that allow the user to monitor the amount of gas remaining. But such gauges are not useful for substances like propane or carbon dioxide,...
-
Compute the volume percent of graphite, VGr, in a 2.5 wt% C cast iron, assuming that all the carbon exists as the graphite phase. Assume densities of 7.9 and 2.3 g/cm3 for ferrite and graphite,...
-
On the basis of microstructure, briefly explain why gray iron is brittle and weak in tension?
-
A cylindrical piece of 8660 steel is to be austenitized and quenched in moderately agitated oil. If the hardness at the surface of the piece must be at least 58 HRC, what is the maximum allowable...
-
A taxpayer expenses out $ 1 5 0 , 0 0 0 of tangible property under IRC Section 1 7 9 when they file their Federal return. The taxpayer will need to apportion the Section 1 7 9 between California...
-
The purpose of this part is to provide you with a better understanding of the origins of the Codification. For this part, you should stay in the Codification, clicking on the tab Other Sources ( on...
-
A ticket is sold for a flat rate of $ 5 8 . 0 0 and the customer pays the $ 5 8 . 0 0 at the time of the sale. What will be the general journal entry for the company selling the ticket at a sales tax...

Study smarter with the SolutionInn App


