Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4
Question:
Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4 in aqueous acid, and (iii) O3, followed by Zn in aceticacid:
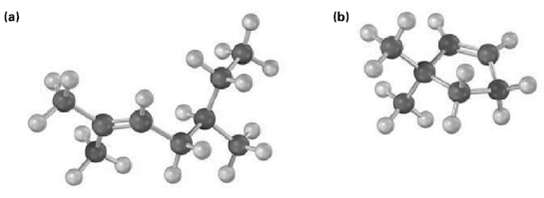
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a CH3 CH3CCHCHCHCHCH3 25Dimethyl2heptene CH3 RCO3H KM...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following alkynes, and predict the products of their reaction with (i) H2 in the presence of a Lindlar catalyst and (ii) H3O+ in the presence ofHgSO4: (b) (a)
-
Predict the products formed by periodic acid cleavage of the following diols. (a) CH3CH(OH)CH(OH)CH3 (b) (c) (d) CHAOH OH OH Ph-CCH OH)CH,CH CH HO HO
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Consider a process consisting of five resources that are operated eight hours per day. The process works on three different products, A, B, and C; Resource Number of Workers Processing Time for A...
-
Sarpedon Corp. claims that its car batteries average at least 880 CCA (cold-cranking amps). Tests on a sample of 9 batteries yield a mean of 871 CCA with a standard deviation of 15.6 CCA. (a) State...
-
The number of vacancies in a material is related to temperature by an Arrhenius equation. If the fraction of lattice points containing vacancies is 8 10-5 at 600 C, determine the fraction of lattice...
-
Use a dot plot to display the data, which represent the weights (in kilograms) of 20 polar bears. Organize the data using the indicated type of graph. Describe any patterns. 426 428 436 545 510 386...
-
Dr. North, a surgeon practicing in Georgia, engaged an Arizona professional corporation consisting of twenty lawyers to represent him in a dispute with a Georgia hospital. West, a member of the law...
-
Task 1: Create a JavaBean Coffee Bean with the following: - Data members: int numSugar; double price; String typeCoffee; Methods: Constructor Accessor Mutator Task 2: Create a HTML CoffeeOrder.html...
-
Access the Call Center Waiting Time file. Each row in the database corresponds to a different call. The column variables are as follows: ProtocolType; indicates protocol type, either PT or PE...
-
One of the chain-termination steps that sometimes occurs to interrupt polymerization is the following reaction between two radicals. Propose a mechanism for the reaction, using fishhook arrows to...
-
Draw the structures of alkenes that would yield the following alcohols on hydration (red = O). Tell in each case whether you would use hydroboration/oxidation oroxymercuration. (b) (a)
-
Darrell Corporation reports under IFRS. At December 31, 2023, the company had a net deferred tax liability of $402,000. An explanation of the items that make up this balance follows: Instructions a....
-
When reviewing a budget comparison report, what can the reader determine?
-
Kevin and Sunny were studying for their mid-term exam on Financial Administration in Sport and discussing financial statements. Kevin stated, "The terminologies used in a balance sheet must be the...
-
Should I include the financial ratio information in this document in the plan to the board of directors?
-
As an economy develops and becomes more integrated into the world economy how do its costs of production change and how well can manage them in both the short-run and long run.
-
Refer to tablel which shows efficient levels of production for a pizzeria that only produces pizza and calzones. What is the opportunity cost of producing one more pizza when the pizzeria moves from...
-
What is the formula for the forward price of an investment asset that provides no income?
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
What happens to the vapor pressure of a substance when its surface area is increased at constant temperature? (a) The vapor pressure increases. (b) The vapor pressure remains the same. (c) The vapor...
-
A student, Flick Flaskflinger, in his twelfth year of graduate work, needed to prepare ethylmagnesium brdmide from ethyl bromide and magnesium, but found that his laboratory was out of diethyl ether....
-
When sec-butylbenzene undergoes free-radical bromi-nation, one major product is formed, If the starting material is optically active, predict whether the substitution product should also be optically...
-
Three alkyl halides, each with the formula C7HBr, have different boiling points. One of the compounds is optically active. Following reaction with Mg in ether, then with water, each compound gives...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


