Name the following alkenes, including the cis or Tran?s designation: (b)
Question:
Name the following alkenes, including the cis or Tran?s designation:
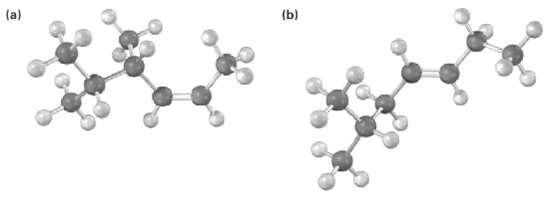
Transcribed Image Text:
(b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
H3 CH3 CH3CHCH CH...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
The name Computer Emergency Response Team is the historic designation for the first team (CERT/CC) at Carnegie Mellon University (CMU). CERT is now a registered service mark of Carnegie Mellon...
-
Suppose the money supply is $250 million dollars and the demand for money is given by Qm D = 400 - 40i, where Qm D is in millions of dollars. A. What is the equilibrium interest rate in this economy?...
-
Which of the four path-goal styles do you think would be the best for managing a group of software engineers? Justify your answer.
-
Prove that the multiplication of two n-digit numbers in base B gives a product of no more than 2n digits.
-
Two sine waves that are out of phase could still have the same two parameters. What are they?
-
1. How should the management team at Bally Hoo make an informed judgment about the future demand for retro clothing styles? 2. What is your evaluation of the creative thinking of Julia? 3. If you...
-
X 3. Ground Water Survey In the following data X = pH of well water Y = Bicarbonate (parts per million) of well water The data is by water well from a random sample of wells in Northwest Texas....
-
Arrange the departments so they satisfy the conditions shown in the following rating grid into a 3 Ã 3 format. Place department 5 in the lower left corner of the 3 Ã 3grid. Department 1...
-
Which of the following compounds can exist as pairs of cisTrans isomers? Draw each cisTrans pair, and indicate the geometry of each isomer. (a) CH3CH = CH2 (b) (CH3)2C = CHCH3 (c) CH3CH2CH = CHCH3...
-
Which member in each of the following sets has higher priority? (a) H or Br (b) C1 or Br (c) CH 3 or CH 2 CH 3 (d) NH 2 or OH (e) CH 2 OH or CH 3 (f) CH 2 OH or CH = O
-
Two charges of equal magnitude Q are held a distance d apart. Consider only points on the line passing through both charges. (a) If the two charges have the same sign, find the location of all points...
-
How can a school protect itself under the first amendment"?
-
Sasha and her family have just returned to Adelaide after living in Latvia for a year. Sasha misses ice skating most of all. She learns that Adelaide has a brand-new ice rink and is excited to...
-
Do you believe contracts should require fewer formalities to be enforceable?
-
Arthur has obtained an order from a human rights tribunal requiring his former employer to pay him damages of $5,000 for discriminating against him in the workplace. To date, Arthur's former employer...
-
How do you schedule a trial? What is the claim you need to file.
-
Without looking back, write down the commonly used form of the accounting equation.
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Nitrogen has a normal boiling point of 77.3 K and a melting point (at 1 atm) of 63.1 K. Its critical temperature is 126.2 K, and its critical pressure is 2.55 * 10 4 torr. It has a triple point at...
-
Draw the structure of 4-isopropyl-2,4,5-trimethylheptane.
-
What is the dissociation constant of an acid that has a pKa of (a) 4 (b) 7.8 (c) -2
-
Using the pka values in Table 3.1, calculate the equilibrium constant for the following reaction. F- acting as a base toward the acid HCN
-
How does the implementation of a centralized database system impact the efficiency and data accuracy in a large retail organization?
-
Identify some processes related to a workplace, assess the benefits and concerns that may arise as a result of the relative sizes of P-time with respect to D-time. In a table include the benefits and...
-
industry competitors and customers analysis & develop a communications pla. Choose any company and analyze its industry, competitors and customers. Then either present theirs communications plan or...

Study smarter with the SolutionInn App


