Predict the major product in each of the following reactions: CH (a) H20 CHCH-CH3H2CH H2SO4 (Addition of
Question:
Predict the major product in each of the following reactions:
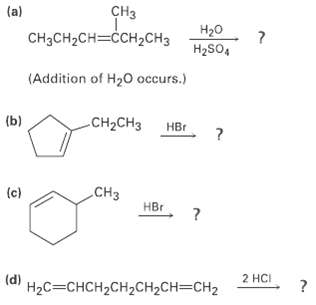
Transcribed Image Text:
CHз (a) H20 CHаCH-CH3ссH2CHз H2SO4 (Addition of H20 occurs.) (b) CH-CHз Нв CHз (c) HBr (d) 2 HCI НаС%3 CHCH2CH2CH2CH%3DCH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
b c CH3 CH3CHCHCCHCH3 CHCH3 HBr CH3 HBr HO HSO...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the major organic product in each of the following reactions:
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Outline the accounting procedures involved in applying the operating method by a lessee.
-
1. In which leadership trait or characteristic does Brittany seem to need the most development? 2. How would you rate Brittany's warmth and enthusiasm as a leader? 3. What action do you recommend...
-
Write the sequence of micro-operations required for the bus structure of Figure 15.6 to add a number to the AC when the number is a. An immediate operand b. A direct-address operand c. An...
-
What does the unit GSE represent on a demodulated spectrum? Is it relevant to other vibration parameters?
-
Financial Accounting) Omar Morena has recently completed his first year of studying accounting. His instructor for next semester has indicated that the primary focus will be the area of financial...
-
Chadwick Enterprises, Incorporated, operates several restaurants throughout the Midwest. Three of its restaurants located in the center of a large urban area have experienced declining profits due to...
-
Solve the following linear recurrences. (a) xk+2 = 2xk - xk+1, where x0 = 1 and x1 = 2. (b) xk+2 = 6xk - xk+1, where x0 = 1 and x1 = 1.
-
The heat of hydrogenation for allene (Problem 6.37) to yield propane is 295kJ/mol, and the heat of hydrogenation for a typical monosubstituted alkene such as propene is 126kJ/mol. Is allene more...
-
Predict the major product from addition of HBr to each of the following alkenes: (a) CH2 (b) (c) CH CH3CHHHH
-
In Exercises, use a derivative routine to obtain the value of the derivative. Give the value to 5 decimal places. f'(1), where f(x) = 1 1+x
-
Technology & Infrastructure Is the current technological system sufficient to implement your proposed plan? Are there any issues with the infrastructure that need to be addressed? How will the data...
-
URVR is a VR file-sharing app that utilizes the 360 camera (or panorama function) on users' phones to capture more than just a snapshot. It captures entire moments, and allows users to share them...
-
Jack is allowed to store his car in a shed in Bert's large car-wrecker yard. Jack has an oral contract only and pays $20 a week. The shed is also used by Bert for storing old batteries and...
-
Now that you've brainstormed ideas, it's time to organize those into an outline. Let's start with a reminder of the prompt: In a well-written paragraph (or two), explain how Dr. Martin Luther King...
-
1. A construction contract called for oak paneling in certain rooms. Because oak was unavailable, a subcontractor installed birch panel. The prime did not notice this deviation. Oak and birch...
-
Enter the following transactions in the accounts: 2016 May 1 Started in business with 18,000 in the bank. 2 Bought goods on time from B. Hind 1,455. 3 Bought goods on time from G. Smart 472. 5 Sold...
-
Where are the olfactory sensory neurons, and why is that site poorly suited for their job?
-
In each pair of compounds, pick the one with the higher boiling point. Explain your reasoning. a. CH 3 OH or CH 3 SH b. CH 3 OCH 3 or CH 3 CH 2 OH c. CH 4 or CH 3 CH 3
-
Using the monomer structure in Table 5.4, draw the structure of poly(vinyl chloride) (PVC), the polymer used for the pipes in household plumbing.
-
Using the monomer structure in Table 5.4, draw the structure of poly(vinyl chloride) (PVC), the polymer used for the pipes in household plumbing.
-
Repeat Problem 5.27 for 1-butene
-
Exactly 24 months ago in 2019, centronics inc issued a 15 year bond. the bond was issued at par to yield 6%. it pays interest annually. over the last 12 months, yields on bonds of similar risk have...
-
How COVID-19 news evoked sentiments and emotions. What are the financial implications of these sentiments for investors/policy makers?
-
How does the network of global stock markets reacts to covid-19? What countries (stock markets) plays most important role during covid-19? Explain.

Study smarter with the SolutionInn App


