Question: Predict the major product that is obtained when the following alkyl halides is treated with potassium terf-butoxide. Explain your reasoning. OCH CH CHI
Predict the major product that is obtained when the following alkyl halides is treated with potassium terf-butoxide. Explain your reasoning.
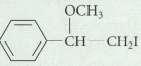
OCH CH CHI
Step by Step Solution
3.30 Rating (165 Votes )
There are 3 Steps involved in it
The benzylic proton is ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
902-O-EE (97).docx
120 KBs Word File


