Predict the product of the following Diels?Alder reaction: ?
Question:
Predict the product of the following Diels?Alder reaction:
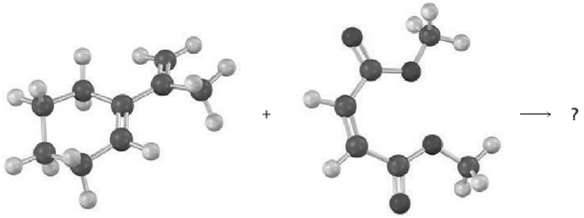
Transcribed Image Text:
?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Rotation of the diene to th...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
135+ Reviews
427+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Predict the product of the following reaction. O (1) BrMg (2) H2O MgBr (1 equiv.)
-
Predict the product of the reaction if CH3-S-CH2CH2CH2CH2-Br is heated in a polar organic solvent, such as methanol. Similarly, what would be the product for hexyl bromide in methanol? (In hexyl...
-
Dixie Irwin is the department manager for Religious Books, a manufacturer of religious books that are sold through Internet companies. Irwins bonus is based on reducing production costs. Irwin has...
-
Why is infrastructure essential to economic development?
-
Obtain the Thevenin equivalent circuit for the circuit in Fig. 13.83 at terminals a-b. j2 52 3 . 10290 V (2 2 4/0 A
-
Use Google Advanced Patent Search to determine the number of skin care-related patents owned by the following cosmetic companies: (a) Avon (b) Kanebo (c) L'Oreal (d) Revlon (e) Shiseido
-
Refer to the Creative Technology example on delaying write-downs of current assets. How much excess inventory do you estimate Creative Technology is holding in March 2005 if the firms optimal days...
-
Dividing LLC Income Martin Farley and Ashley Clark formed a limited liability company with an operating agreement that provided a salary allowance of $56,000 and $45,000 to each member, respectively....
-
1. Create and upload a histogram of the salary data for the city of Bell, where each bar width is about 50,000 US dollars. (Data for the histogram is at the bottom). a.) Is the distribution of the...
-
Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate tos-cis? (a) (c) (b)
-
Draw a segment of the polymer that might be prepared from 2-phenyl-1, 3-butadiene.
-
Show that, if (1 and (2 satisfy the time-dependent Schrdinger equation, then c1(1 + c2(2 satisfies this equation, where c1 and c2 are constants.
-
Assume you are a media planner and you have just completed a media plan using both magazine and television advertising. Your magazine schedule reaches 50 percent of the target audience and your...
-
UXZ is a wedding event organiser company based in Z - land. The Finance Manager at UXZ would like to use activity - based costing principles to apply the correct overhead costs to the three wedding...
-
The distribution of natural log of family income in the US in 2017 was Normal with mean 11.237 and standard deviation 0.747, according to data from the St. Louis Fed. (a) (2 points) What is the...
-
A plan is under consideration to open a large tract of the Amazon rainforest in Brazil to commercial mining and logging operations on territory several indigenous cultures consider to be their...
-
Read below and explain two different types of taxes that may be affected by their death and whether those taxes can be eliminated through planning (California Laws Apply) FAMILY DATA and Facts...
-
Find the inverse Laplace transform by using the partialfraction expansion method. \(\frac{s+10}{s\left(s^{2}+2 s+5 ight)}\)
-
Consider the following cash flows in Table P5.5. (a) Calculate the payback period for each project. (b) Determine whether it is meaningful to calculate a payback period for project D. (c) Assuming...
-
What is the effect on vapor pressure of a solution with particularly strong solutesolvent interactions? With particularly weak solutesolvent interactions?
-
(a) Use the polygon rule to draw an energy diagram (as in Figures 16-5 and 16-7) for the MOs of a planar cyclooctatetraenyl system. In Figure 16.5 In Figure 16.7 (b) Fill in the eight pi electrons...
-
Step 2 of the iodination of benzene shows water acting as a base and removing a proton from the sigma complex. We did not consider the possibility of water acting as a nucleophile and attacking the...
-
In an aqueous solution containing sodium bicarbonate, aniline reacts quickly with bromine to give 2, 4, 6-tribromoaniline. Nitration of aniline requires very strong conditions, however, and the...
-
Show how binary exponentiation can be used to calculate a 128 efficiently. How many multiplications are required?
-
How can one tell many months the positive skew had in it?
-
Drug 4 mg I every 3 hours prn pain. Supplied as 10mg/10 ml. Calculate mL per dose?

Study smarter with the SolutionInn App


