Predict the product(s) of the following reactions: CH (b) CH-CH (a) 1. DIBAH 2. H30+ 1. CH2CH,MgBr
Question:
Predict the product(s) of the following reactions:
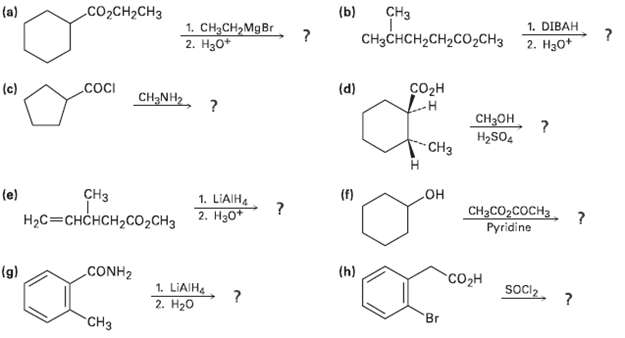
Transcribed Image Text:
CHз (b) СооCH-CHз (a) 1. DIBAH 2. H30+ 1. CH2CH,MgBr 2. Нао" CHзснCH-CH2CO2CH3 (d) Созн .cocI (c) CH NH2, ? CH3он H2SO4 "CHз но CH3CO2COCH3 (f) СНз 1. LIAIH (e) 2. Нзо" Pyridine НаС3 СHCHCH>со,сна (h) "сон CONH2 (g) Socl2. 1. LIAIH. 2. H20 Br "CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b c O0 OCHCH3 1 CH3CHMgBr 2 H30 CH3 CH3CHCHCHCOCH 3 i The 1 DIBA...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of each of the following Diels-Alder reactions for the diene (left) and dienophile (right) as shown below. 0 0 0 0
-
Predict whether the following reactions will be spontaneous in acidic solution under standard conditions: (a) Oxidation of Sn to Sn2+ by I2 (to form I-) (b) Reduction of Ni2+ to Ni by I- (to form I2)...
-
Predict whether the following reactions would occur spontaneously in aqueous solution at 25C. Assume that the initial concentrations of dissolved species are all 1.0 M. (a) Ca(s) + Cd2+(aq) Ca2+(aq)...
-
A man drops a stone from a high bridge and hears it strike the water below exactly 4 s later. (a) Estimate the distance to the water based on the assumption that the travel time for the sound to...
-
Why should even practiced speakers plan their presentations when addressing a business audience instead of just "winging it"?
-
Two rooftop cisterns supply a tropical bungalow with shower water. The water surface in the uppermost cistern (A) is 8 m above the ground, and the water surface in the lower cistern (B) is 7 m above...
-
The numbers of deaths caused by fire per year from 1990 to 2005 in New South Wales Find the range, mean, variance, and standard deviation of the population data set. 8 13 2 11 4 2 2 4 4 3 5 5 14 1 6...
-
1. Assume you are the CEO of Peerless Products and you are aware of your companys lack of end-to-end supply chain capability. What are some of the high-level adverse impacts on your business that may...
-
The net income of the Donald and Browning partnership is $451000. The partnership agreement specifies that Donald and Browning have a salary allowance of $116000 and $189000, respectively. The...
-
1. Using the data, develop a level production plan. How much extra cost (inventory and HR costs) are involved in this plan? What additional costs (both financial and nonfinancial) might be involved...
-
How might you prepare the following compounds from butanoic acid? (a) 1-Bulanol (b) Butanal (c) 1-Bromobutane (d) Pentanenitrile (e) 1-Butene (f) N-Methylpentanamidc (g) 2-Hexanone (h) Butyl benzene...
-
Predict the product, if any, of reaction between propanoyl chloride and the following reagents: (a) Li (Ph) 2 Cu in ether (b) LiA1H 4 , then H 3 O + (c) CH 3 MgBr, then H 3 O + (d) H 3 O + (e)...
-
Use the remainder to find a bound on the error in approximating the following quantities with the nth-order Taylor polynomial centered at 0. Estimates are not unique. ln 1.04, n = 3
-
Briefly comment and point out something interesting from the following post: As Mr. Peters, I would first evaluate the exchange rate risk by analyzing historical trends and economic factors impacting...
-
What are electrical engineering formulas, and why are they crucial in the field? What is Ohm's Law, and how is it expressed in formula form?
-
Thorprods, Inc., common stock is listed on the New York Stock Exchange and satisfies the listing requirements of Form S-3, but it is not a well-known seasoned issuer. Thorprods common shares...
-
Outline three reasons why a company may choose to develop a business plan. 2 . Outline the key steps involved in developing a business plan. 3. Outline five common components of a business plan. 4 ....
-
The sale of travel insurance is governed by the Financial Services Reform Act 2001. Based on the requirements of this Act, you must supply specific documentation to the customer. What information...
-
Number of cars owned Classify the variable as qualitative or quantitative.
-
A spacecraft has left the earth and is moving toward Mars. An observer on the earth finds that, relative to measurements made when the spacecraft was at rest, its a. length is shorter b. KE is less...
-
The valence electron configurations of several atoms are shown here. How many bonds can each atom make without hybridization? a. Be 2s 2 b. P 3s 2 3p 3 c. F 2s 2 2p 5
-
The 60-MHz proton NMR spectrum of 2, 2, 3, 3-tetra-chlorobutane consists of a sharp singlet at 25oC, but at -45oC consists of two singlets of different intensities separated by about 10 Hz. Explain...
-
The 60-MHz proton NMR spectrum of 2, 2, 3, 3-tetra-chlorobutane consists of a sharp singlet at 25oC, but at -45oC consists of two singlets of different intensities separated by about 10 Hz. Explain...
-
What changes would you expect in the 13C NMR spectrum of 1-bromopropane upon cooling the compound to very low temperature?
-
A horizontal board of negligible thickness and area 4.0 m hangs from a spring scale that reads 60 N when a 2.0 m/s wind moves below the board. The air above the board is stationary. When the wind...
-
The only force acting on a 2.2 kg body as it moves along the positive x axis has an x component Fx = -5x N, where x is in meters. The velocity of the body at x = 2.5 m is 9.0 m/s. (a) What is the...
-
Use conservation of energy to determine the angular speed of the spool shown in the figure below after the 3.00-kg bucket has fallen 3.70 m, starting from rest. The light string attached to the...

Study smarter with the SolutionInn App


