Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH
Question:
Predict the products of the following reactions:
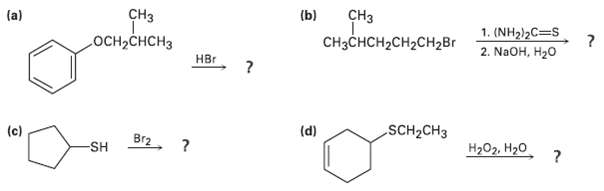
Transcribed Image Text:
CHз (b) CHз (a) 1. (NH2)2C=S 2. NaOH, H20 осн-снCHз CнзснсH2CH2CH2Br Hвr SCH2CH3 (d) (c) Br2, ? Hа0z. Н2о SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
b O d CH3 OCHCHCH3 CH3 CH3...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
To load and register a JDBC driver, two popular methods can be used: either using the Class.forName() method or creating a new instance of the Driver class.(True/False)
-
What issues do they encounter when attempting to measure the economy?
-
In the same study described in Exercise 8.19, the sodium content was also measured for the sampled hamburgers, and the sample mean and standard deviation were 658 and 47 milligrams, respectively....
-
Explain how and why the method of instrumental variables allows us to estimate the labor demand elasticity.
-
Using the information for Vision, Inc., in SE4 and SE5, compute the debt to equity ratio, return on equity, and the interest coverage ratio for 2013 and 2014. In 2012 total stockholders equity was...
-
On Monday, April 5, 2010, just before 3:00 in the afternoon, miners at Massey Energy Corporation's Upper Big Branch coal mine in southern West Virginia were in the process of a routine shift change....
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
How would you prepare the following compounds from 1-phenylethanol? (a) Methyl 1-phenyl ethyl ether (b) Phenylepoxyethane (c) tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How have social media like Twitter and Facebook changed marketing communications?
-
Does the hedonic treadmill help or hinder the pursuit of happiness? You must explore both the pros and cons here. From or class material, or any other source, give reasons why?
-
How long will it take a Taka 10,000 investment to double if it earns 7 percent annual interest. (b) Assume that you have taken a loan of Taka 25,000 at the rate of 10% for three years. Using the...
-
Mallory is very excited as she has just found the perfect condo! She feels confident that the bank will provide her with a mortgage as she received a pre-approval certificate. Mallory has worked hard...
-
Russell received a $300 refund of his 2022 State income taxes in May of 2023. If Russell took the standard deduction in 2022, how much of the $300 refund must be included as income in 2023?
-
Explain the necessary components of an executive summary and share the city and only one recommendation that you have proposed to expand and grow by building a new hospital. Describe two components...
-
Which proposed allocation in Exhibit 1 would be most appropriate for the Fund given its characteristics? A. Allocation 1 B. Allocation 2 C. Allocation 3 William Azarov is a portfolio manager for...
-
Q:1 Take any product or service offered in Pakistan and apply all determinents of customer Perceived value ?
-
It is necessary to estimate how rapidly a piece of equipment can be evacuated. The equipment, which is 0.7 m 3 in volume, initially contains carbon dioxide at 340 K and 1 bar pressure. The equipment...
-
Assign the proper formal charge to the colored atom in each of the following structures: (a) (b) (c) (d) (e) (f) (g) (h) H-C-H H-C-NH H-C-H
-
Write a condensed structural formula for the following compound.
-
Write each of the following condensed structural formulas as a bond-line formula: (a) (CH3)2CHCH2CH3 (b) (CH3)2CHCH2CH2OH (c) (CH3)2C==CHCH2CH3 (d) CH3CH2CH2CH2CH3 (e) CH3CH2CH(OH)CH2CH3 (f) CH2...
-
You have a business worth $1,000,000 and other assets (including your home) worth an additional $750,000. What is the recommended automobile insurance policy that you should carry to be FULLY...
-
Republic Services and Waste Management company Compare the companies' long term Revenue, Gross Profit, Operating Profit) and Net Income and related growth trends. What is the average three year...
-
A young couple own a home in NJ. They would like to have Homeowner Insurance coverage for their home in NJ, in case of frozen plumping and damage form heavy snow or power surges. Which Homeowner's...

Study smarter with the SolutionInn App


