Predict the product(s) of the following reactions. If more than one product is formed, tell which ismajor.
Question:
Predict the product(s) of the following reactions. If more than one product is formed, tell which ismajor.
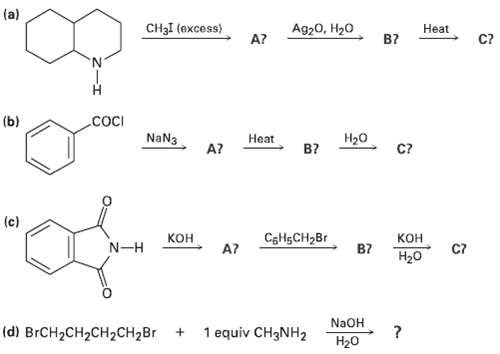
Transcribed Image Text:
(a) CH3I (excess) Ag20, H20 Нeat C? A? B? .cocI (b) H20 NaN3 Нeat C? B? A? (c) Кон CgHsCH2Br КОн C7 N-H A? B7 Нао NaOH (d) BRCH2CH2CH2CH2BR 1 equiv CH3NH2 H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
N H H3C CH3 minor CH3I excess H3C CH3 H3C CH3 major ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
If more than one computer can update a replicated database, what three problems can occur?
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the product of each of the following Diels-Alder reactions for the diene (left) and dienophile (right) as shown below. 0 0 0 0
-
Calculate the new profit sharing ratio after the admission of Fischer. Assignment Ubisoft Entertainment is a partnership of exclusive game distributors who specialize in electronic arts and play...
-
Why do you think it is important to tell employee the purpose of a policy?
-
Use the following two equations: (1) P = 12 2Q (2) P = 3 +Q a. Find the equilibrium price and quantity. b. Graph the demand and supply equations. Illustrate the equilibrium point.
-
Suppose that the current measurements in a strip of wire are assumed to follow a normal distribution with a mean of 10 milliamperes and a variance of 4 (milliamperes) \({ }^{2}\). What is the...
-
CbSSports.com developed the Total Player Ratings system to rate players in the National basketball Association (NbA) based upon various offensive and defensive statistics. The following data show the...
-
1. (10 points) We want to approximate the function ln x for x = [1, e] using a polynomial to the precision of 10-8. How should we choose the lowest degree polynomial to minimize the error over the...
-
Three years ago, the Oak Street TCBY bought a frozen yogurt machine for $8,000. A salesman has just suggested to the TCBY manager that she replace the machine with a new, $12,500 machine. The manager...
-
What are the major products you would expect from Hofmann elimination of the following amines?
-
Fill in the missing reagents a?c in the following scheme: NH2 CHCH3 CH=CH2 CCH3 b, c CHCH2NCH3 CH CH-CH2
-
Repeat Exercise 2 using the Runge-Kutta method of order four. In Exercise 2 a. y' = e ty , 0 t 1, y(0) = 1, with h = 0.5; actual solution y(t) = ln(e t + e 1). b. y' = (1 + t)/(1 + y), 1 t 2, y(1)...
-
what ways do trust-based exchanges contribute to economic efficiency and social cohesion, facilitating cooperation, collaboration, and value creation within and across organizational boundaries?
-
Complete the mechanism for the deprotonation of the given ketone by potassium tert-butoxide (KO'Bu). Complete the curved arrows for the first step, and draw the structure of the enolate intermediate...
-
How does the concept of change leadership differ from traditional notions of management, and what competencies are required for leaders to effectively navigate the complexities of change, inspire...
-
13. A heavy rope, 40 ft long, weighs 0.4 lb/ft and hangs over the edge of a building 110 ft high. How much work is done in pulling the rope to the top of the building? How much work is required to...
-
Complete the following reaction schemes by adding the appropriate reactant(s), reagent(s), conditions, and/or major product(s). Make sure to indicate the proper regiochemistry and stereochemistry...
-
What is the matching principle and what types of accounts are matched according to this principle. What is the outcome for a business?
-
A company produces earbuds. The revenue from the sale of x units of these earbuds is R = 8x. The cost to produce x units of earbuds is C = 3x + 1500. In what interval will the company at least break...
-
The three diagrams represent three different solutions of the binary acid HA. Water molecules have been omitted for clarity, and hydronium ions (H 3 O + ) are represented by hydrogen ions (H + )....
-
Draw all possible staggered and eclipsed conformations of 1-bromo-2-chloroethane (see the following balland- stick model), using Newman projections. Underneath each structure, draw the corresponding...
-
Draw the formula for the preferred conformation of a. Cis-1,4-dimethylcyclohexane b. Trans-1-isopropyl-3-methylcyclohexane c. 1,1-diethylcyclopentane d. Ethylcyclohexane
-
Name the following cis-trans pairs: a. b. Cl Cl Cl Cl Br Br CH3 CH3
-
What percentage of the American opportunity tax credit may be refundable?
-
You want to buy a house. You have $50,000 saved for the down payment and can pay $2,000 per month on the mortgage. The 30-year mortgage interest rate is 9% per year (equal to 0.75% per month). What...
-
Compute the Social Security and Medicare for Paul, Alex, and Andrea. The rates are 6 . 2 % for Social Security and 1 . 4 5 % for Medicare. Assume the Social Security limit is $ 1 4 2 , 8 0 0 and the...

Study smarter with the SolutionInn App


