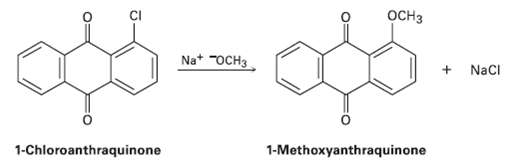
Propose a mechanism for the reaction of 1-chioroanthraquinone with methoxide ion to give the substitution product 1-methoxyanthraquinone.
Question:
Propose a mechanism for the reaction of 1-chioroanthraquinone with methoxide ion to give the substitution product 1-methoxyanthraquinone. Use curved arrows to show the electron flow in eachstep.
Transcribed Image Text:
OCH3 Na* "OCH3 Naci 1-Chloroanthraquinone 1-Methoxyanthraquinone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
0 CI OCH3 co Addition of the nucleophile OCH3 The carb...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the reaction of phenyl isocyanate with ethanol.
-
(a) Propose a mechanism for the reaction of benzyl alcohol with acetyl chloride to give benzyl acetate. (b) Propose a mechanism for the reaction of benzoic acid with acetyl chloride to give acetic...
-
What is the result of the following? A. 0 B. 0.007 C. The code does not compile due to line 7. D. The code does not compile due to line 8. E. The code does not compile for another reason. 1: import...
-
During 2016, Inez had the following transactions involving capital assets: Gain on the sale of unimproved land (held as an investment for 3 years)......................$ 3,000 Loss on the sale of a...
-
Determine the ending balance of each of the following T-accounts. a. b. c. d. e. f. Cash 50 100 300 60 20 Accounts Payable 2,000 8,000 2,700
-
Names over Time The website http://www.visualcinnamon.com/babynamesus gives a spaghetti plot showing the popularity of the top 10 baby names for each year 1880 to 2014 (use the window scroller at the...
-
I recently sold my used car. If no new production occurred for this transaction, how could it have created value?
-
The Field, Brown & Snow are partners and share income and losses equality. The partner decide to liquidate the partnership when their capital balances are as follows: Field, $131,100; Brown,...
-
solve x+x-x+5=0 x+2x-4=0 z-2z+1=0
-
Draw resonance structures of the intermediate carbocations in the bromination of naphthalene, and account for the fact that naphthalene undergoes electrophilic substitution at C1 rather than C2. Br...
-
4-Chloropyridine undergoes reaction with dim ethylamine to yield 4-dimethylaminopyridine. Propose a mechanism for thereaction. CI N(CH3)2 HN(CH3)2 HCI N.
-
a. Estimate beta for each of the following securities assuming that the standard deviation of returns for the market portfolio (m) is 8.0 percent. b. Based on the Capital Asset Pricing Model, with a...
-
Holtzman Clothiers's stock currently sells for $39.00 a share. It just paid a dividend of $1.25 a share (i.e., Do = $1.25). The dividend is expected to grow at a constant rate of 4% a year. 1-What...
-
Answering Required: Introduction of process of budgetary control project Create the plan of project QUESTION Below are the steps in the Process of Budgetary Control. Choose a project you had from...
-
A European-style bond carries a 7% coupon rate, a face value of $1,000, and has 10 years until it matures. If the bond is currently trading at a price of $1200, what is the nominal yield to maturity...
-
A CMO has three tranches. Tranche A has a principal of $40.94 million at an annual coupon of 7.69%. Tranche B has a principal of $36.47 million at an annual coupon of 9.43%. Tranche Z has a principal...
-
A new three-year CMO has two tranches. The 'A' tranche has a principal of $59.5 million with an annual-coupon of 5.41%. The 'Z' tranche has a coupon of 7.37% with a principal of $67.1 million. The...
-
Contrast the common-law rule allowing a person to resist an unlawful arrest with the modern trend of requiring citizens to submit to unlawful arrests by police officers. Which approach makes more...
-
President Lee Coone has asked you to continue planning for an integrated corporate NDAS network. Ultimately, this network will link all the offices with the Tampa head office and become the...
-
A Rankine power generation cycle is operated with water as the working fluid. It is found that 100 MW of power is produced in the turbine by 89 kg/s of steam that enters the turbine at 700 C and 5...
-
Explain which of the three products shown in is formed when 1-butene reacts with HCI.
-
Explain which of the four products shown in is formed when cis-2-pentene reacts with CI2 andwater.
-
Explain which of the four products shown in is formed when cyclopentene reacts with CI2 andwater.
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)
-
How many monthly withdrawals of $1,400 will an investment of $75,000 sustain if the first withdrawal is made 12 months from now and the money earns 8.4% compounded monthly
-
1. You buy a bond with 3 years left to maturity and a yield to maturity of 6% for $920. After 1 year you receive a coupon payment of $30 and sell the bond for $940. What was your rate of return on...

Study smarter with the SolutionInn App


