Rank the following compounds in order of increasingacidity: (a) CH3CH2CO2H (b) CH3CH2OH (c) (CH3CH2)2NH (d) CH3COCH3 (e)
Question:
Rank the following compounds in order of increasingacidity:
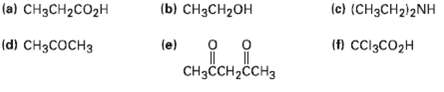
Transcribed Image Text:
(a) CH3CH2CO2H (b) CH3CH2OH (c) (CH3CH2)2NH (d) CH3COCH3 (e) (f) CCI3CO2H CнзСCH-CCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Check your answer by using Tab...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following compounds in order of increasing stability based on relative ring strain.
-
Rank the following compounds in order of decreasing acidity:
-
Rank the following compounds in order of decreasing nucleophilicity: a. b. c. H2O and NH3 in methanol d. Br-, Cl-, I- in methanol CH,CO, CHCH2S, CH3CH20 in methanol
-
Prepare journal entries to record the following grant-related transactions of an enterprise fund activity. Explain how these transactions should be reported in the enterprise funds financial...
-
What motivating impulse may prompt individuals to agree to requests that do not directly benefit themselves or their organizations?
-
Determine the force in each member of the roof truss. State if the members are in tension or compression. Assume all members are pin connected. 3 m E- |B |C 10 kN 10 kN 10 kN -4 m-4m-4 m-4 m-
-
What are some reasons a potential prospect might not be readily accessible? How far should you go to try to overcome such an accessibility problem before you move to the next lead?
-
Listed below are the eight activity cost pools used by Charvez Corporation. Production set-up costs Heating costs Machinery power costs Purchasing department costs Maintenance costs Design and...
-
Hemingway Company purchases equipment by issuing a 7-year, $280,000 non-interest-bearing note, when the market rate for this type of note is 8%. Hemingway will pay off the note with equal payments to...
-
Baker Industries net income is $24,000, its interest expense is $5,000, and its tax rate is 40%. Its notes payable equals $27,000, long-term debt equals $75,000, and common equity equals $250,000....
-
Identify all the acidic hydrogen?s (pKa (b) (c) (a) CHCH2H3 CH H-CH23H C02CH (f) COCI (d) (e) CHCH2CC 3CH2 CH CH2CN
-
Write resonance structures for the followinganions: (a) (b) {c) -, N=CCHCOCH3 CHH CH3CH (e) (d) H OCH3
-
Using the data for Problem 16.15 on page 625 that represent U.S. total computer and software sales (in $mil-lions) from 1992 through 2011 (stored in Computer Sales), a. Fit a third-order...
-
Step #1: Brainstorm your purpose Purpose = talents + passions + skills/expertise + values Identify at least 4 attributes for each area of your personal and professional talents, passions,...
-
discuss the role of non-coding RNAs, such as microRNAs, long non-coding RNAs, and small interfering RNAs, in post-transcriptional gene regulation, chromatin remodeling, and epigenetic silencing, and...
-
- 1 y=x+e- H find the derivatives 6. y=xln (x) cos x
-
Why will learning about organizational behavior help a person be a better employee? Give an example of how a person could apply the information about organizational behavior to their job (or a...
-
Suppose Johnson & Johnson and the Walgreen Company have the expected returns and volatilities shown below, with a correlation of 22.6%. Johnson & Johnson Walgreen Company E[R] 7.9% 10.7% SD [R] 16.4%...
-
A television weather forecaster once said that there was a 50 percent chance of rain on Saturday and a 50 percent chance of rain on Sunday, and therefore a 100 percent chance of rain that weekend....
-
Does log 81 (2401) = log 3 (7)? Verify the claim algebraically.
-
In valence bond theory, what determines the geometry of a molecule?
-
Draw structures analogous to those in Eqs. 18.59a-d for the catalytic intermediates formed in the conversion of 1, 7-octadiene to cyclohexene and ethylene catalyzed by the G2 catalyst. Eqs. 18.59a-d...
-
Within series, arrange the compounds according to increasing rates of their reactions by the SN1 - E1 mechanism. Explain your reasoning. CH CH Br
-
Which of the, two phenols in each set is more acidic? Explain. Phenol or m-chlorophenol
-
In what ways does HR assist managers in handling employee performance issues or conflicts within their teams? What role does HR play in helping managers navigate challenges related to allegations and...
-
Define the following sets (30 points) (a) X = {(x, y) = R: y = f(x) = 3x + 3y 42} =1 (b) Y={(x, y) = R: y = f(x) = 2x + 2y 42} (c) Z = XUY (d) Prove whether or not each set defined above is convex?
-
Why is A/B testing so useful for marketing, and digital marketing in particular? Give at least two examples of A/B testing for marketing purposes. Give a null and alternative hypothesis for one of...

Study smarter with the SolutionInn App


