Show the conjugate acid of each of these species: a) NH d) CHC=C: b) :0: -N: c)
Question:
Show the conjugate acid of each of these species:
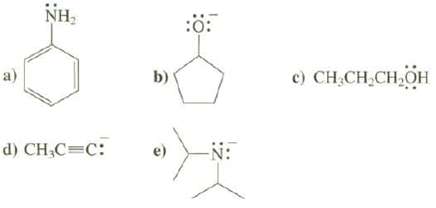
Transcribed Image Text:
a) NH₂ d) CH₂C=C: b) :0: -N: c) CH₂CH₂CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
To get the conjugate acid ...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
a. Draw the conjugate acid of each of the following: (1) NH3 (2) Cl- (3) HO- (4) H2O b. Draw the conjugate base of each of the following: (1) NH3 (2) HBr (3) HNO3 (4) H2O
-
What is the conjugate acid of each of the following bases? (a) HSO4- (b) H2O (c) CH3NH2 (d) -NH2 (e) CH3CH2 (f) CH3CO2-
-
Write the formula for the conjugate acid of each of the following bases: (a) HS-, (b) HCO3-, (c) CO32-, (d) H2PO4-, (e) HPO42-, (f) PO43-, (g) HSO4-, (h) SO42-, (i) SO32-.
-
The labor force participation rate is increasing in Japan because... a) there are fewer unemployed people b) the working age population is increasing c) retirement ages are increasing d) more women...
-
Green Ltd produces a special valve used in the burners of gas stoves. The firm uses the first-in, first-out (FIFO) process costing method for product costing. The costs entered into work in process...
-
Gold Door Credit Bank is offering 9.3 percent compounded daily on its savings accounts. If you deposit $4,500 today, how much will you have in the account in 5 years? In 10 years? In 20 years?
-
What is the difference between the operations of a class and its attributes?
-
On February 1, 2011, Charo Mendez purchased 6% bonds issued by CR Utilities at a cost of $30,000, which is their par value. The bonds pay interest semiannually on July 31 and January 31. For 2011,...
-
Assume an investor buys a call option on XYZ stock with a strike price of $80 for $6 when the stock is trading for $77. What is the investor's break-even stock price?
-
Consider the portion of an excavator shown. At the instant under consideration, the hydraulic cylinder is extending at a rate of 6 in. /sec, which is decreasing at the rate of 2 in. /sec every...
-
Explain whether each of the following solvents would be acceptable for reactions involving this anion: (a) Liquid NH3 (b) CH3CH2OH (c)CH3CH2OCH2CH3 CHC=C: solvent
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
At what frequency is the reactance of a 10-F capacitor (a) 1 , (b) 100 , and (c) 0.01 ?
-
Kenya Corporation had an equity structure that consisted of $1 par value common stock, $3,500,000; paid-in capital in excess of par, $17,500,000; and retained earnings, $22,700,000. (a) Assuming that...
-
Consider an economy with a single traded asset and three possible states of the world (i.e., \(N=1\) and \(S=3\) ), with dividend \(D=(0.5,1,2)^{\top}\) and price \(p=1\). For the payoff...
-
Stearns Corporation was a diversified company with two separate lines of business - chemicals and financial services. At the beginning of 20X7, Strearns sold its financial services unit, resulting in...
-
Consider an economy populated by \(I\) agents with exponential utility functions of the form \[u^{i}(x)=-\frac{1}{a_{i}} \mathrm{e}^{-a_{i} x}, \quad \text { with } a_{i}>0 \quad \text { for all }...
-
The transfer function of a dynamic system is given by \[G(s)=\frac{20 s+50}{s^{3}+10 s^{2}-5 s-30}\] a. Using Routh's stability criterion, determine the stability of the open-loop system. b. Suppose...
-
In March 1990, Bill Foley, research manager for Herman Miller, Inc., began a routine evaluation of new woods to use in the firm's signature piece--the \$2,277 (the 1990 cost) Eames chair. The Eames...
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
Evaluate (2i + 2'). i=1
-
Where would you expect each of the following compounds to absorb in the IR spectrum? (a) 4-Penten-2-one (b) 3-Penten-2-one (c) 2, 2-Dimethylcyclopentanone (d) m-Chloro benzaldehyde (e)...
-
How might you use mass spectrometry to distinguish between the following pairs of isomers? (a) 3-Methyl-2-hexanone and 4-methyl-2-hexanone (b) 3-Heptanone and 4-heptanone (c) 2-Methylpentanal and...
-
Tell the prominent IR absorptions and mass spectral peaks you would expect for the following compound:
-
It is possible to form an atom out of a muon (like and electron, but with mass approximately given by 105.7 MeV/c 2 ). What would the muonic Bohr radius be? Give a numerical value, and make sure your...
-
Suppose you drop little balls of mass m on to a target marked on the floor. According to the uncertainty principle, the balls should not necessarily fall straight down even if you attempt to hold...
-
First , explain what Social Exchange Theory is and how it functions in Interpersonal Relationships. Second , describe a situation (real or hypothetical) where Social Exchange Theory is a factor in a...

Study smarter with the SolutionInn App


