Show the products you would obtain by reduction of the following esters withLiAlH4: (b) (a)
Question:
Show the products you would obtain by reduction of the following esters withLiAlH4:
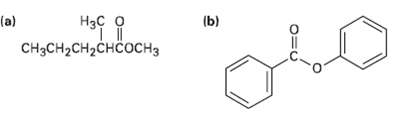
Transcribed Image Text:
(b) Нас о (a) CHясH2CH2снсоснз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (20 reviews)
Lithium aluminum hydride reduces an est...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products you would obtain by acid-catalyzed reaction of Cyclohexanone with ethylamine, CH3CH2NH2, and with diethyl amine, (CH3CH2)2NH.
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
The following data represent the concentration of dissolved organic carbon (mg/L) collected from 20 samples of organic soil. Assume that the population is normally distributed. Complete parts (a)...
-
Why is searching for a job both exhilarating and intimidating? How can you overcome feelings of intimidation?
-
She sees an eagle with probability 0.2 during an hour of observation, a jackrabbit with probability 0.5, and both with probability 0.05. An ecologist is looking for the effects of eagle predation on...
-
You are the lead systems developer for your local university and have been told that the athletic department would like to modernize their online ticket sales and ordering system for sporting events....
-
Going Places, Inc., manufactures a variety of luggage for airline passengers. The company has several luggage production divisions, including the Suitable Cases Division, as well as a wholly owned...
-
1 Acts of civil disobedience, especially in the 1960's, were used to protest Unjust Laws. Who gets to decide what "unjust" is? Is it unjust just because thousands or more agree they don't like the...
-
Jet Spray Corp, now part of IMI Cornelius, manufactures dispensers for beverages. One of Jet Spray's best-known products is a two-product cold beverage dispenser, the twin Jet 3 (TJ3), which is made...
-
What product would you expect from the reaction of butyrolactone with LiAlH4 with DIBAH? Butyrolactone
-
What ester an what Grignard reagent might you start with to prepare the following alcohols? (b) , H (c) HCH-H2H2,H CH-CH (a)
-
What is the role of an actuary relative to pension plans what are actuarial assumptions?
-
Exercise 9 - 4 A ( Algo ) Inventory turnover LO 0 9 - 0 2 Selected financial information for Solomon Company for Year 4 follows: \ table [ [ Sales , $ 2 , 3 5 0 , 0 0 0 es Exercise 9-4A (Algo)...
-
1 . Today, you can buy a 1 5 - year Treasury bond with a 3 . 5 % coupon rate for $ 1 0 4 7 . 5 5 . Show your work. A ) Calculate the Yield - to - Maturity for this bond B ) Assume the bond is...
-
What major risk to retirement do annuities mitigate? Why is that important?
-
QUESTION 1 - 1 POINT For a Sunday dinner party, each guest can choose from the 7 different kinds of food to eat. If any number of food types can be chosen, how many food type combinations are...
-
Milner Company is working on two job orders. The job cost sheets show the following. Job 201 Job 202 Direct materials $7,500 $9,250 Direct labor 4,000 8,150 Manufacturing overhead 5,050 9,600 Prepare...
-
A rock deep in Earth has a temperature higher than the melting point it has at Earths surface. Why can this buried rock remain in the solid state even though its temperature is very high?
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
Using the molecular orbital energy ordering for second-row homonuclear diatomic molecules in which the 2p orbitals lie at lower energy than the 2p draw MO energy diagrams and predict the bond order...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
What is the wavelength of light with a wave-number of 1720 cm-1?
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
5. Consider the 'top-hat' function which is zero everywhere except between -1 and 1 where it takes value 1. Show, using only elementary integration, that the convolution of this function with itself...
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...

Study smarter with the SolutionInn App


