Question: Show the structure of the carbocation that is observed when this compound is dissolved in superacid. CH3
Show the structure of the carbocation that is observed when this compound is dissolved in superacid.
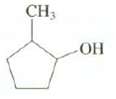
CH3
Step by Step Solution
3.40 Rating (162 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-N-R (47).docx
120 KBs Word File


