Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic
Question:
Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic solution:
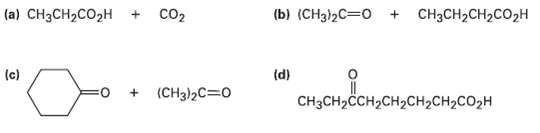
Transcribed Image Text:
(b) (CHз])2С—о + |(a) CHзCH2CO2н co2 CH3CH2CH2CO2H (d) (c) (CH3)2C=0 CнзCH-CCH2CH2CHCH2CO2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Remember that alkenes can give ketones carboxylic a...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In each of the following pairs, determine whether the two represent resonance forms of a single species or depict different substances. If two structures are not resonance forms, explain why. All the...
-
Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. H (b) CH CH3 /Pd CH2H2H2CH H2/Pd (d) CH Br CHH2CHCH (c) Br2 HCI ,...
-
Show the structures of all possible acid-catalyzed dehydration products of the following. If more than one alkene is possible, predict which one will be formed in the largest amount. a....
-
Consider the frequency demodulation scheme in Figure in which the incoming FM signal s (t) is passed through a delay line that produces a phase-shift of ?/2 radians at the carrier frequency ? c . The...
-
Given P(A) = .40, P(B) = .50, and P(A B) = .05. (a) Find P(A | B). (b) In this problem, are A and B independent?
-
A cyclical load of 1500 lb is to be exerted at the end of a 10-in.-long aluminum beam (Figure 7-17). The bar must survive for at least 106 cycles. What is the minimum diameter of the bar?
-
The numbers of earned degrees conferred (in thousands) in 2014 are shown in the table at the right. Use a pie chart to organize the data. Earned Degrees Conferred in 2014 Type of Number degree (in...
-
Roberta Santos, age 41, is single and lives at 120 Sanborne Avenue, Springfield, IL 60781. Her Social Security number is 123-45-6789. Roberta has been divorced from her former husband, Wayne, for...
-
A steam turbine having an isentropic turbine efficiency of 90% drives an air compressor having an isentropic compressor efficiency of 85%. Steady-state operating data are provided on the figure....
-
11. Let x [n] be real WSS with correlation r [m] which is assumed known. The second order prediction is formulated as follows. Define e [n] via: e [n] = x [n] + ax [n 1] + ax [n- 2] We want to find...
-
Draw the structure of an alkene that yields only acetone, (CH3)2C = O, on ozonolysis followed by treatment with Zn.
-
Compound A has the formula C10H16. On catalytic hydrogenation over palladium, it reacts with only I molar equivalent of H2. Compound A also undergoes reaction with ozone, followed by zinc treatment,...
-
Can a shock wave develop in the converging section of a converging-diverging nozzle? Explain.
-
explaining how having an incarcerated parents has affected you, your parents, and other members of your family
-
Projects are chosen to be part of an organizational portfolio based on their ability to help the organization achieve its goals. Once chosen, it is important that projects are successful, or an...
-
What are the types of revenue sources used by states in the U.S. for funding schools?
-
Consider the following data. Period Rate of Return (%) 1 -6.0 2 -8.0 3 -4.0 4 2.0 5 5.4 What is the mean growth rate over these five periods?
-
If you were conducting an actuarial valuation for an Australian defined benefit superannuation fund, what assumption for salary (wage) inflation would you use? In particular, consider a fund whose...
-
How much does someone with a short contract gain or lose when the three-month Eurodollar futures price changes from 97.540 to 98.080?
-
Would you use the adjacency matrix structure or the adjacency list structure in each of the following cases? Justify your choice. a. The graph has 10,000 vertices and 20,000 edges, and it is...
-
What are the main properties of liquids (in contrast to gases and solids)?
-
Choose the alkyl halide(s) frorn the following list of C6H13Br isomers that meet each criterion below. (1) l-bromohexane (2) 3 -bromo-3 -methylpentane (3 ) I -bromo-2,2-dimethylbutane (4) 3 -bromo-2-...
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
The following situation is an example for a fire detection and suppression system in an office building. The Initiating Event (IE) for the Event Tree is "fire starts", with a frequency of once every...
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
Evaluate the use of novel catalytic systems, such as structured catalysts, catalytic distillation, and multifunctional catalysts, in achieving process intensification, discussing the effects on...

Study smarter with the SolutionInn App


