Question: Show that where is the anlge between u and F. KK = 1 (u2/c2) cos2 1-u/c F
Show that where is the anlge between u and F.
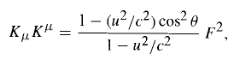
KK" = 1 (u2/c2) cos2 1-u/c F
Step by Step Solution
3.37 Rating (172 Votes )
There are 3 Steps involved in it
KK K KK From Eq 1270 K K 1 dE 1 d cdr 1u c1uc ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
5-P-E-E-R (39).docx
120 KBs Word File


