Some radio-nuclides decay by capturing one of their own atomic electrons, K- shell electrons, say. An example
Question:
Some radio-nuclides decay by capturing one of their own atomic electrons, K- shell electrons, say. An example is below. Show that the disintegration energy Q for this process is given by, where mV and mTi, arte the atomic masses of 49V and 49Ti, respectively, and EK is the binding energy of the vanadium K-shell electron.
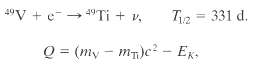
Transcribed Image Text:
4°V + e-- 49TI + v. = 331 d. T2 Q = (my – mT)c? - Ex.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Assuming the neutrino has negligible mass then Amc mmymc Now since Vanadium has 23 electrons see A...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Modern Physics questions
-
Show that the disintegration energy for decay is Q = (mp mD me)c2 = (Mp MD)c2, where the ms represent the masses of the parent and daughter nuclei and the Ms represent the masses of the neutral...
-
Show that the disintegration energy for decay is Q = (mp mD me)c2 = (Mp MD 2me)c2, where the ms represent the masses of the parent and daughter nuclei and the Ms represent the masses of the...
-
Find the disintegration energy Q for the decay of 49V by K-electron capture. The needed data are mv = 48.948 52u, mTi = 48.947 87 u, and Ek = 5.47keV.
-
Its estimated that the average corporate user sends and receives some 112 e-mails daily.78 Thats about 14 e-mails per hour, and even if half of those dont require a lot of time and concentration,...
-
Wilson Marketing Consulting, a proprietorship owned by Lin Wilson, was started on January 1, 2013, with an investment of $50,000 cash. The company prepares marketing plans for clients. It has been...
-
To remove the stain from the dress, Rob treated the stain with a cleaner containing the hydrolytic enzyme amylase. Explain what happens to starch at the molecular level when it is acted upon by...
-
The data in Table 19.5 originate from the 1991 edition of the Historical Loss Development Study, published by the Reinsurance Association of American. These data have been widely used to illustrate...
-
Vidi Corporation made the following purchases related to its property, plant, and equipment during its fiscal year ended December 31, 2014. The company uses the straight-line method of depreciation...
-
If f is any function from A into B, we can describe the inverse image as a function from B into P(A), which is also commonly denoted . If b B, (b) = {a A | f(a) = b}. If does have an inverse, the...
-
1, Assuming that VMD MIC competitors have an accurate costing system, what are the characteristics of jobs (services) that VMD MIC is likely to win vs. lose to their competitors? Provide a graph...
-
The cesium isotope 137Cs is present in the fallout from aboveground detonations of nuclear bombs. Because it decays with a slow (30.2y) half-life into 137Ba, releasing considerable energy in the...
-
The radionuclide 11 C decays according to 11 C 11 B + e + + v, T 1/2 = 20.3. The maximum energy of the positions is 0.960MeV. (a) Show that the designation energy Q for this process is given by Q =...
-
Steel It began January with 55 units of iron inventory that cost $35 each. During January, the company completed the following inventory transactions: Requirements 1. Prepare a perpetual inventory...
-
Date Information 13/05/20XX BAC Commercial pays monthly rent of $2000 by EFT. 15/05/20XX The tenant (renter) Jill Sharp pays by EFT the rental of $1350 for 15 Drysdale Ave. 20/05/20XX Fred Sykes...
-
How will attending the DVAA benefit your agency or department as a whole?
-
Use the above information to complete the following tasks: Prepare a cash budget for the months of January 2022, February 2022, March 2022 and April 2022. Based on the cash budget, provide...
-
What happens if an organization does not have the three sections listed on their statement of cash flows?
-
You decided to create a new language! Your computer memory has 4 bits, so what is the Maximum number of symbols you can encode in this memory?
-
Let \(\left(B_{t}ight)_{t \geqslant 0}\) be a \(\mathrm{BM}^{1}\). Decide which of the following processes are Brownian motions: a) \(X_{t}:=2 B_{t / 4}\); b) \(Y_{t}:=B_{2 t}-B_{t}\); c)...
-
Without solving, determine the character of the solutions of each equation in the complex number system. 3x 2 3x + 4 = 0
-
A certain DAC has the following specifications: eight-bit resolution, full scale = 2.55 V, offset 2 mV; accuracy = 0.1% F.S. A static test on this DAC produces the results shown in Table 11-11. What...
-
Two long straight wires with equal cross-sectional radii a are located parallel to each other in air. The distance between their axes equals b. Find the mutual capacitance of the wires per unit...
-
A long straight wire is located parallel to an infinite conducting plate. The wire cross-sectional radius is equal to a, the distance between the axis of the wire and the plane equals b. Find the...
-
Find the capacitance of a system of two identical metal balls of radius a if the distance between their centers is equal to b, with b >> a. The system is located in a uniform dielectric with...
-
Given the following Python code: x = 20 while x > 10: #do something x = x 1 How many times this loop will iterate. 10 11 20 20 0
-
The HTML program is fine I want help to figure out what's wrong with CSS that would not make my website look the same as the picture I provided. @charset "UTF-8"; /* CSS Document for CA3 */ body {...
-
Consider a twisted pair link of distance 2 km. It is required to compute the amount of received power Pr, assuming the transmit power Pt = 1 Watt and the cable attenuation is 20 dB/km.

Study smarter with the SolutionInn App


