Compare the effect of the Efron-Morris estimator on the baseball data in Section 8.3 with the effect
Question:
Compare the effect of the Efron-Morris estimator on the baseball data in Section 8.3 with the effect of a James-Stein estimator which shrinks the values of πi towards π0 = 0.25 or equivalently shrinks the values of Xi towards µ, = 2√n sin-1 √π0.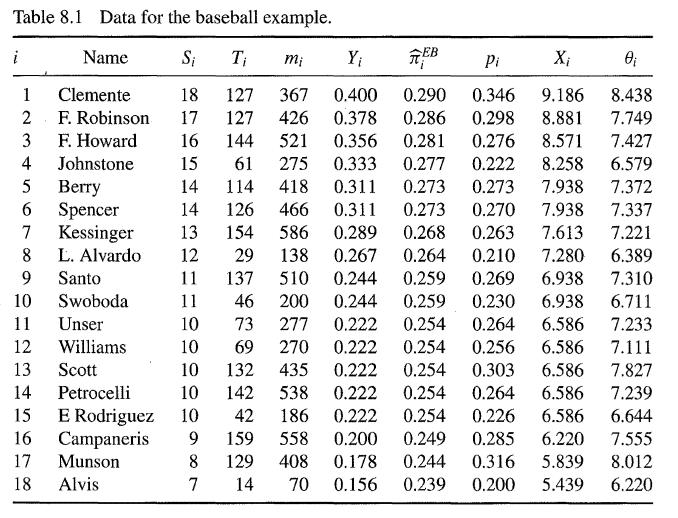
Transcribed Image Text:
Table 8.1 Data for the baseball example. Name Si Ti mi Y₁ FEB Pi X₁ 0₁ 18 127 367 8.438 0.400 0.290 0.346 9.186 0.298 8.881 7.749 16 144 17 127 426 0.378 0.286 521 0.356 275 0.333 0.277 0.281 0.276 8.571 7.427 15 61 0.222 8.258 6.579 14 114 418 0.311 0.273 0.273 7.938 7.372 14 126 466 0.311 0.273 0.270 7.938 7.337 13 154 586 0.289 0.268 0.263 7.613 7.221 L. Alvardo 12 29 138 0.267 0.264 0.210 7.280 6.389 11 137 510 0.244 0.259 0.269 6.938 7.310 11 46 200 0.244 0.259 0.230 6.938 6.711 10 73 277 0.222 0.254 0.264 6.586 7.233 10 69 270 0.222 0.254 0.256 6.586 7.111 132 435 0.222 0.254 0.303 6.586 142 538 0.222 0.254 0.264 6.586 10 42 186 0.222 0.254 0.226 6.586 6.644 9 159 558 0.200 0.249 0.285 6.220 7.555 8 129 408 0.178 0.244 0.316 5.839 8.012 7 14 70 0.156 0.239 0.200 5.439 6.220 i 1 2 3 4 5 6 7 8 9 10 11 Clemente F. Robinson F. Howard Johnstone Berry Spencer Kessinger Santo Swoboda Unser Williams 12 13 Scott 14 Petrocelli 15 E Rodriguez 16 Campaneris 17 Munson 18 Alvis 10 10 7.827 7.239
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
Brandy Corporation, a calendar-year, accrual-basis taxpayer, signed a contract on December 27, 20X1, for the installation of a new central air conditioning unit. Because of various shipping delays,...
-
For the following exercises, determine why the function f is discontinuous at a given point a on the graph. State which condition fails. [3+x, f(x) = {x, x, x <1 x = 1 a = 1 x> 1
-
A study was designed to compare the effect of a vitamin E supplement on the growth of guinea pigs. There were 15 guinea pigs available for the study. The guinea pigs were randomly assigned to one of...
-
The following table includes transactions carried out by Ram Horn Corporation, as well as columns for each of the three categories found in the statement of cash flows: operating, financing, and...
-
The following events occurred after the end of the companys fiscal year but before the annual audit was completed. Classify each event as to its impact on the financial statements, that is, (1)...
-
Dyna Distribution markets CDs of the performing artist King James. At the beginning of March, Dyna had in beginning inventory 1,500 King James CDs with a unit cost of $7. During March, Dyna made the...
-
Founded in 1966 as a trader of oil and oil products, Vitol is a company with no external shareholders. All shareholders are also employees. It is a conglomerate company of energy companies that work...
-
Environ Company has determined that its new fireplace screen would gain widespread customer acceptance if the company could price it at or under $90. Anticipated labor hours and costs for each unit...
-
Is discord supported by a relational database, hierarchical database, NoSQL database, or something else?Explain
-
The Helmert transformation is defined by the matrix so that the element a ij in row i, column j is It is also useful to write a j for the ( column) vector which consists of the jth column of the...
-
Carry out the Bayesian analysis for known overall mean developed in Section 8.2 mentioned earlier (a) with the loss function replaced by a weighted mean and (b) with it replaced by 2(0,6) = ;; - 03, -
-
Lithium carbonate solution reacts with aqueous hydrobromic acid to give a solution of lithium bromide, carbon dioxide gas, and water. Write the molecular equation and the net ionic equation for the...
-
Wildcat Company is considering a 5-for-3 stock split. The current price is $57.00 per share, and Wildcat believes that its total market value would increase by 7.5% as a result of the improved...
-
What are the evolutionary origins and diversification of enzyme families, superfamilies, and functional motifs across different taxa and biochemical pathways, and how do sequence-structure-function...
-
Your company wants to purchase 10,000 crawfish from a supplier. One supplier demands a payment of $100,000 today plus $10 per crawfish payable in one year. Another supplier will charge $21 per...
-
A restaurant has a variable cost percentage of 42%, a fixed cost percentage of 20%, and a profit percentage of 10%. When the menu planner uses the actual pricing method for a dish with a food cost...
-
You purchased shares in Arrow Electronics (ticker: ARW) 3mo ago at a price of $90.00 / share. During the time you owned the shares, they paid no quarterly dividends. Today, the shares are worth...
-
Yousuf Company began operations in 2013 and determined its ending inventory at cost and at lower-of-cost-or-market at December 31, 2013, and December 31, 2014. This information is presented below....
-
Catalytic hydrogenation of naphthalene over PdC results in rapid addition of 2 moles of H 2 . Propose a structure for this product.
-
What promotion blend would be most appropriate for producers of the following established products? Assume average- to large-sized firms in each case and support your answer. a. Chocolate candy bars....
-
A small company has developed an innovative new spray-on glass cleaner that prevents the buildup of electrostatic dust on computer screens and TVs. Give examples of some low-cost ways the firm might...
-
Would promotion be successful in expanding the general demand for: ( a ) almonds, ( b ) air travel, ( c ) golf clubs, ( d ) walking shoes, ( e ) high-octane unleaded gasoline, ( f ) single-serving,...
-
Investigate some applications of exponential and logarithmic functions in science and engineering. Tell us why you think it is interesting. Discuss a topic different from the ones mentioned already...
-
The heat of vaporization AH, of dichloromethane (CH2Cl2) is 28.0 kJ/mol. Calculate the change in entropy AS when 280. g of dichloromethane condenses at 39.8 C. Be sure your answer contains a unit...
-
Evaluate the integral, ez dz - z(z i)(z 3) - where is the circle of radius 2 centered at the origin. Hint: use Cauchy's theorem or Cauchy's integral formula.

Study smarter with the SolutionInn App


