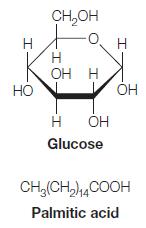
On page 351 we showed that the oxidation of glucose and palmitic acid yields 15.94 kJ/g and
Question:
On page 351 we showed that the oxidation of glucose and palmitic acid yields 15.94 kJ/g and 38.90 kJ/g, respectively. Calculate these values in terms of kJ/mol and kJ per carbon atom oxidized for both glucose and palmitic acid.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:





