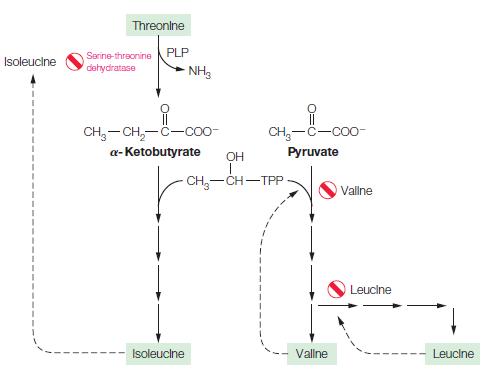
Propose a mechanism for the TPP-dependent step in isoleucine biosynthesis (see Figure 18.20). Draw the predicted product
Question:
Propose a mechanism for the TPP-dependent step in isoleucine biosynthesis (see Figure 18.20). Draw the predicted product of this reaction.
Figure 18.20.
Transcribed Image Text:
Isoleucine Threonine Serine-threonine PLP dehydratase NH3 CH-CH₂-C-coo a-Ketobutyrate OH Isoleucine CH₂-C-COO- Pyruvate -CH₂-CH-TPP Vallne Vallne Leucine Leucine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The image you provided illustrates a portion of the amino acid biosynthesis pathways for isoleucine ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for the reaction of benzyl acetate with methylamine. Label the attacking nucleophile and the leaving group, and draw the transition state in which the leaving group leaves.
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Measuring the height of a California redwood tree is very difficult because these trees grow to heights over 300 feet. People familiar with these trees understand that the height of a California...
-
Refer to information provided in E7-9 as you answer the following questions. 1. If a manager for Howard Company wanted to query the companys database to obtain information about customer sales during...
-
Suppose that in Problem 6-15, only a one-half fraction of the 2 4 design could be run. Construct the design and perform the analysis, using the data from replicate I. Problem 6-15. A nickel-titanium...
-
The response variable \(Y_{i j}\) in a \(2^{2}\) design can also be expressed as a regression model \[Y_{i j}=\mu+\beta_{1} x_{1}+\beta_{2} x_{2}+\beta_{12} x_{1} x_{2}+\varepsilon_{i j}\] where the...
-
Tofias Corporation recently hired a new accountant with extensive experience in accounting for partnerships. Because of the pressure of the new job, the accountant was unable to review what he had...
-
Amounts are in thousands of dollars ( except number of shares and price per share ) : Kiwi Fruit Company Balance Sheet Cash and equivalents $ 3 7 0 Operating assets 7 8 0 Property, plant, and...
-
Why is phenylketonuria resulting from dihydropteridine reductase deficiency a more serious disorder than PKU resulting from phenylalanine hydroxylase deficiency?
-
Most bacterial mutants that require isoleucine for growth also require valine. Why? Which enzyme or reaction would be defective in a mutant requiring only isoleucine (not valine) for growth?
-
Have a student identify countries-of-origin of popular U.S. cars. Find out how many cars that we identify as American are made in other countries and how many cars we identify as foreign are...
-
I need someone to verify the numbers I have on Form 1120 are correct based on the Excel Data provided below. All the data and information that I have for this is provided below. I was supposed to...
-
1. Gas phase decomposition of di-t-butyl peroxide is monitored in a batch reactor of constant volume at isothermal conditions of 170C. (CH3)3COOC(CH3)3 C2H6+2 CH3COCH3 The run is started with pure...
-
Find the the following in Maybelin product. - Regulations & Labour laws - Misleading Advertisements - Unionized Employees - Lawsuits for aircraft failure - Safety Standards - Quality Standards
-
In the picture below there are four point charges, each q1= q2= +5e, q3 =+3e, q4 = -12e, e = electron. Distance d = 5 m. Determine: a. The electric field intensity at point P of the four charges b....
-
Make a simpe java code that asks if you are 18+ using arrays. MAKE SURE TO USE ARRAYS the question could be " Are you 18?" type accept or decline and if they hit accept connect to this code:...
-
There are various types of accounting changes requiring different types of reporting treatments. Understanding the different changes is important to analysis of financial statements. Required: a....
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
Why does the enzyme reaction for chymotrypsin proceed in two phases?
-
List three proteases and their substrates.
-
What is the relationship between the action of salicylate or aspirin that researchers believe accounts for some of its therapeutic effects?
-
Saturn's moon Kiviuq was discovered by a Canadian-led team in 2000. It has a semimajor axis of 11,000,000 km. What is its orbital period around Saturn, in Earth days?
-
1. How is heat transfer related to temperature? Describe a situation in which heat transfer occurs. What are the resulting forms of energy? When heat transfers into a system, is the energy stored as...
-
The space age bicycle rim in Figure 4.88 can be modeled as a thin disk with four circular cutouts, which are of identi- cal size. The density of the disk material is kg/m2 . In Figure 4.88, the rim...

Study smarter with the SolutionInn App


