Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the
Question:
Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the ideal-gas process shown in Figure Q 17.9. Then show the process as a first-law bar chart.
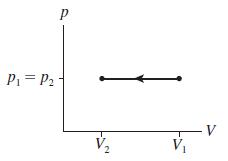
Figure Q 17.9
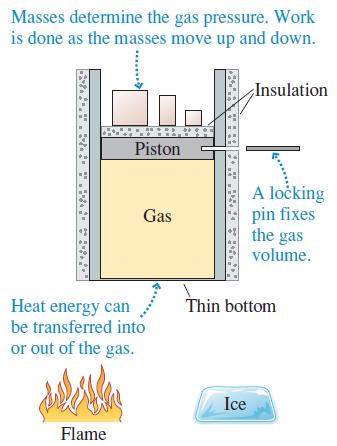
Figure Q 17.13
Transcribed Image Text:
P1= P2 V V2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
This is a case of gas compression and therefore is a po...View the full answer

Answered By

Milan Mondal
I am milan mondal have done my Msc in physics (special astrophysics and relativity) from the University of burdwan and Bed in physical science from the same University.
From 2018 I am working as pgt physics teacher in kendriya vidyalaya no2 kharagpur ,west bengal. And also I am doing advanced physics expert in chegg.com .also I teach Bsc physics .
I love to teach physics and acience.
If you give me a chance I will give my best to you.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Identify three situations in which you use self-service delivery. For each situation, what is your motivation for using self-service delivery, rather than having service personnel do it for you?
-
Consider a borrow-and-invest strategy in which you use $1 million of your own money and borrow another $1 million (at the t-bill rate) to invest $2 million in a market index fund. If the risk free...
-
In a cylinder filled up with ideal gas and closed from both ends there is a piston of mass m and cross-sectional area S (Fig. 4.21). In equilibrium the piston divides the cylinder into two equal...
-
A restaurant records the following data over a month for its food: Opening inventory: $31,000 Purchases: $88,000 Closing Inventory: $28,000 Transfers in: $800 Transfers out: $200 Employee meals:...
-
A refrigerator removes heat from a refrigerated space at 0oC at a rate of 2.2kJ/s and rejects it to an environment at 20oC. The minimum required power input is (a) 89 W (b) 150 W (c) 161 W (d) 557 W...
-
An engineer is studying the effect of cutting speed on the rate of metal removal in a machining operation. However, the rate of metal removal is also related to the hardness of the test specimen....
-
In a domestic refrigerator, the refrigerant commonly used is (a) Air (b) Carbon dioxide (c) Ammonia (d) Freon-12
-
Multiple Choice 1. What is a basic premise of the acquisition method regarding accounting for a noncontrolling interest? a. Consolidated financial statements should be primarily for the benefit of...
-
How would you facilitate your own town hall meeting if you were an administrator at Francis Howell School?
-
In your final project, you will assume the role of an accountant and complete the year-end adjustment process for your company using a provided workbook. This workbook is the first deliverable (Part...
-
A compressed-air cylinder is known to fail if the pressure exceeds 110 atm. A cylinder that was filled to 25 atm at 20C is stored in a warehouse. Unfortunately, the warehouse catches fire and the...
-
Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the ideal-gas process shown in Figure Q 17.10. Then show the process as a first-law bar chart. Figure Q 17.10...
-
Contrast the process and the requirements of proof required in a civil as opposed to a criminal action.
-
Germanys $95 billion in revenue electronics giant, Siemens AG, sells medical and telecommunications equipment, power plants, automotive products, and computers. Siemens has been operating in the...
-
Suppose England raised its corporate tax rate by one percentage point. How would this increase affect the economics of the IDC-U.K. project?
-
Apnex, Inc., is a biotechnology firm that is about to announce the results of its clinical trials of a potential new cancer drug. If the trials are successful, Apnex stock would be worth $62 per...
-
On March 28, 2019, Netflix had an equity beta of 1.56 calculated over the last three years using monthly returns on Netflix and the S&P 500 as the market proxy. The S&P 500 and Netflix had a standard...
-
In mid-2012, Ralston Purina had AA-rated, 10-year bonds outstanding with a yield to maturity of 1.73%. a. What is the highest expected return these bonds could have? b. At the time, similar maturity...
-
Multiple-Concept Example 10 reviews most of the concepts that are needed to solve this problem. Material found with a mummy in the arid highlands of southern Peru has a 14 6 C activity per gram of...
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
Two blocks are attached to opposite ends of a massless rope that goes over a massless, frictionless, stationary pulley. One of the blocks, with a mass of 6.0 kg, accelerates downward at 3/4 g. What...
-
Two blocks are attached to opposite ends of a massless rope that goes over a massless, frictionless, stationary pulley. One of the blocks, with a mass of 6.0 kg, accelerates downward at 3/4 g. What...
-
The cable cars in San Francisco are pulled along their tracks by an underground steel cable that moves along at 9.5 mph. The cable is driven by large motors at a central power station and extends,...
-
A project requires an initial investment of $100,000 to purchase equipment. The equipment will be depreciated on a straight-line basis over the five-year life of the project. The company expects the...
-
Vodaphone's earnings per share in 2022 was $1.80, and in 2017 it was $1.25. The company distributes 60% of its earnings as dividends, and the stock is currently valued at $37.75. The cost to issue...
-
A bond is currently selling in the market for $1,085.96. It has a coupon of 8% and a 15-year maturity. Using annual compounding, what is the yield to maturity on this bond?

Study smarter with the SolutionInn App


