Does each of the configurations in Figure Q 41.6 represent a possible electron configuration of an element?
Question:
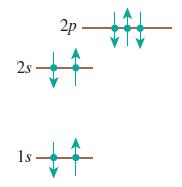
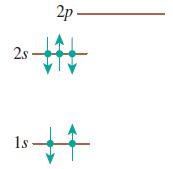
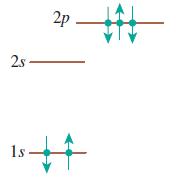
Does each of the configurations in Figure Q 41.6 represent a possible electron configuration of an element? If so, (i) identify the element and (ii) determine whether this is the ground state or an excited state. If not, why not?
(a)
(b)
(c)
Transcribed Image Text:
2p - %23 2s- %2. 1s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Yes this is the ground state of nitrogen see Figure 4122 Note that the p subshells have ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Determine the current /for each of the configurations of Fig. 2.150 using the approximate equivalent model for the diode. In Figure 2.150 (a) (b) (c) 10 Si 2V 20 V Si 10 20 Si - 10V 10 Si
-
In an underwriting, what role does each of the following play? (a) The investment banker (b) The syndicate (c) The red herring (d) The SEC (e) The saver (investor).
-
Does each of the following items increase or decrease the cost of goods sold? (a) Freight in, (b) Beginning inventory, (c) Purchases discounts, (d) Ending inventory
-
With only a straightedge and compass, use a number line and the Pythagorean theorem to construct a segment whose length is 2. Measure the segment as accurately as possible, and write your answer in...
-
A researcher is interested in the relationship between the time (measured in minutes) spent exercising per week and scores on a life satisfaction scale that ranges from 10 to 70, with higher scores...
-
Blue Company incurred the following expenditures to buy a new machine: Invoice, $28,000 less 9% cash discount. Freight charges, $490. Assembly charges, $1,100. Special base to support machine,...
-
Verify the entries in Table 13.8 for the gamma distribution. Specifically: a. Show that the gamma is a member of the linear exponential family of distributions. b. Describe the components of the...
-
Can anyone be trained to be a project manager?
-
Convert the following NFA to an equivalent deterministic finite automaton: Start 0.1 Provide both the state diagram as well as the definition of each component of the proposed M = (E,Q, S, A, 6).
-
A soldier jumps from a plane and opens his parachute when his velocity reaches the terminal velocity V T . The parachute slows him down to his landing velocity of V F . After the parachute is...
-
When all quantum numbers are considered, how many different quantum states are there for a hydrogen atom with n = 1? With n = 2 With n = 3? List the quantum numbers of each state.
-
What are E and L (as a multiple of h) of a hydrogen atom in the 6f state?
-
The following asset is not considered a financial investment under IFRS: a . Trading securities. b . Associate investment. c . Held-for-collection securities. d . Inventories.
-
Today is April 12, 2024, and you, CPA, are a senior accountant with Kenney & Niu LLP. You just got out of a meeting with a new client, Alan Morneau, who has approached your firm for some tax advice....
-
Ghostbuster's is considering replacing their proton packLinks to an external site. weapons that are presently used to eliminate paranormal pests. Old Weapons New Weapons Original cost $50,000 $70,000...
-
Mr. Lupez asks about services that he received at the beginning of the year. He states that the insurance company did not pay anything towards those charges, and he is wondering why. You explain that...
-
A monthly depositor has accumulated $110,000 in fica and federal income taxes in their payable account as of July. 31. Which of the follow would be correct? Group of answer choices No deposit is...
-
Keeper Ltd. has 50,000 no par value common shares authorized, issued, and outstanding. All 50,000 shares were issued at $4 per share. Retained earnings are $60,000. If 5,000 common shares were...
-
A major drug store chain wishes to build a new warehouse to serve the whole Midwest. At the moment, it is looking at three possible locations. The factors, weights, and ratings being considered are...
-
What types of questions can be answered by analyzing financial statements?
-
An object whose moment of inertia is 4.0 kg m 2 experiences the torque shown in Figure EX12.25. What is the objects angular velocity at t = 3.0 s? Assume it starts from rest. (Nm) 0+ -t (s) 3 FIGURE...
-
An object whose moment of inertia is 4.0 kg m 2 experiences the torque shown in Figure EX12.25. What is the objects angular velocity at t = 3.0 s? Assume it starts from rest. (Nm) 0+ -t (s) 3 FIGURE...
-
A 1.0 kg ball and a 2.0 kg ball are connected by a 1.0-m-long rigid, massless rod. The rod is rotating cw about its center of mass at 20 rpm. What net torque will bring the balls to a halt in 5.0 s?
-
MR Green is one of the resident for who you take care in a nursing home .He is recovering from a stroke and has a pressure ulcer . In addition he is disoriented and confuse . However he can feed...
-
What does the accountability of prescriptive theory? b. What does the accountability of capture theory? c. What does the accountability of normative accounting theory? d, What does the accountability...
-
Describe the connections between leader accountability and how leaders hold people accountable for their actions? sustainability through accountability.

Study smarter with the SolutionInn App


