a. Name and give the formulae of substances A to E. b. Write balanced chemical equations for
Question:
a. Name and give the formulae of substances A to E.
b. Write balanced chemical equations for the reactions in which compounds B, C and E were formed.
c. Where would you expect to find acid A?
d. Universal indicator solution was added to solution C. What colour did it go?
e. Upon addition of dilute hydrochloric acid to solution C, a neutralisation reaction took place.
(i) Write a balanced chemical equation for the reaction taking place.
(ii) Name the salt produced in this reaction.
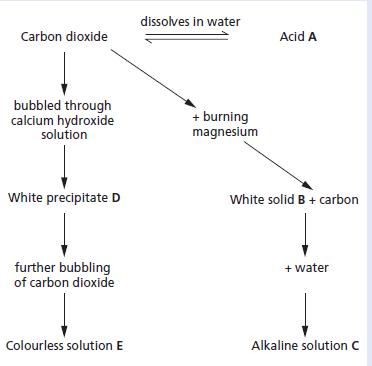
dissolves in water Carbon dioxide Acid A bubbled through calcium hydroxide solution + burning magnesium White precipitate D White solid B + carbon further bubbling of carbon dioxide + water Colourless solution E Alkaline solution C
Step by Step Answer:
a b A HCO3 C MgOH2 E CaHCO32 for B CO g Mg s for C ...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
In what chemical shift ranges would you expect to find the proton NMR signals of ethyl acetate (CH3CO2CH2CH3)?
-
What kind of organizational structure would you expect to find in (a) A fast-food restaurant (b) A company like GE or GM (c) A biotechnology company?
-
Would you expect to find adenineguanine or cytosinethymine base pairs in DNA? Why?
-
Factor each polynomial. 64y 9 + z 6
-
A solution was prepared by dissolving 0.834 g of sulfur, S8, in 100.0 g of acetic acid, HC2H3O2. Calculate the freezing point and boiling point of the solution.
-
Lynsar Corporation started as a single plant to produce its major components and then assembled its main product into electric motors. Lynsar later expanded by developing outside markets for some...
-
Draw a graph similar to the one shown in exhibit 13.4 and explain its implications. Exhibit 13.4 EXHIBIT 13.4 Bayside Memorial Hospital: Corporate and Project Costs of Capital Project Cost of Capital...
-
Favorite Foods Corp. sold its food to stores and distributors. It established a quantity discount scale that was publicly published and made available to all buyers. The top of the scale gave the...
-
11. A planet is revolving around the sun in an elliptical orbit. The mass of planet is m, angular momentum of planet about sun is L, and length of semi major axis is a and eccentricity are e. Time...
-
1. Why do you think Rolls has continued to bear this structural currency mismatch so long? Why hasn't it done what many automobile companies have done, and move some of their manufacturing and...
-
Limestone is an important raw material used in many different industries. a. One of the properties of limestone is that it reacts with acids. (i) Why do farmers spread powdered limestone on their...
-
The following question is about carbon dioxide. a. Name and give the formula of each of the substances A, B and C. b. Identify by name the different pieces of apparatus D, E, F and G. c. Draw and...
-
How does a company usually decide if an IS or IT function should be outsourced? If they are operating effectively? If people are using them to their best advantage?
-
Immigration Law: why do immigration laws change so constantly? What is the last update made and what's your opinion on it?
-
You expect to receive $ 1 0 , 0 0 0 at the end of year 1 , $ 5 , 0 0 0 at the end of year 2 and $ 4 , 0 0 0 at the end of year 3 . If your required rate of return is 8 % , what is the uneven cash...
-
Smith manufactures 1,991 electronic pianos and reports the following per unit costs related to a particular part. Direct materials: $2.0 Direct labor: $3 Variable OH: $1 Fixed OH: $5 An outside...
-
One of the main human resource issues that Amazon faces is Employee Well-being and Work-Life Balance . The demanding nature of Amazon's operations and high-performance expectations can lead to...
-
It has been said that International Law remains a " contradiction in terms ." Discuss how International Law treats individual persons (i.e. human beings) compared to nation states ? Explain your...
-
Indicate whether each statement is always true or sometimes false. Justify your answer by giving a logical argument or a counterexample. (a) A square matrix with linearly independent column vectors...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
A photon-powered spacecraft of mass 10.0 kg emits radiation of wavelength 225 nm with a power of 1.50 kW entirely in the backward direction. To what speed will it have accelerated after 10.0 y if...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
The work function for metallic cesium is 2.14 eV. Calculate the kinetic energy and the speed of the electrons ejected by light of wavelength (a) 700 nm, (b) 300 nm.
-
A car lease requires payments of $445 at the beginning of each month for 6 years. If the lease rate is 4.30% compounded monthly, what should be the selling price of the car if you can purchase the...
-
Anthony's investment manager offers him an interest rate of 8.00% compounded monthly on his investments. How much more money would he have to deposit at the end of every month for his fund to...
-
Grandpa wants to give you $580 at the beginning of each month while you are at school for 3 years. If interest is 4.7% compounded monthly, how much does she have to invest today to provide this?

Study smarter with the SolutionInn App


