Carbon-fibre-reinforced plastic (CRP) is used in the manufacture of golf clubs and tennis rackets. a. What are
Question:
Carbon-fibre-reinforced plastic (CRP) is used in the manufacture of golf clubs and tennis rackets.
a. What are composite materials?
b. Which two substances are used to manufacture this composite material? Consider the data in the table.
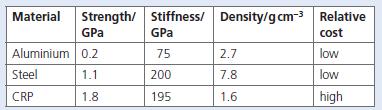
c. Discuss the advantages and disadvantages of using the three materials above in the manufacture of golf clubs.
Transcribed Image Text:
Stiffness/ Density/gcm-3 Relative GPa Material Strength/ GPa cost Aluminium 0.2 75 2.7 low Steel 1.1 200 7.8 low CRP 1.8 195 1.6 high
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
a Com pos ite materials are materials composed of two or more different substances The different sub...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The head box process is used in the manufacture of paper to transform the pulp slurry flow into a jet of 2 cm and then spread it onto a mesh belt [22]. To achieve desirable paper quality, the pulp...
-
A press produces parts used in the manufacture of large-screen plasma televisions. If the press is correctly adjusted, it produces parts with a scrap rate of 5%. If it is not adjusted correctly, it...
-
Carbon tetrachloride is a colorless liquid used in the manufacture of fluorocarbons and as an industrial solvent. How many molecules are there in 7.58 mg of carbon tetrachloride?
-
Redo Exercise 29 for the situation in which Ms. Jones withdrew $1000 at the end of the seventh year instead of depositing it. Data in Exercise 29 Ms. Jones deposited $100 at the end of each month for...
-
Give the conjugate acid to each of the following species regarded as bases. a. HSe b. NH2 c. ClO2 d. N2H4
-
How would a Kantian thinker respond to the Pavlik case?
-
Your firm uses a large parallel-plate capacitor to store energy, and you measure the electric field strength between the plates to determine the amount of energy stored. During a test run with a new...
-
Two independent companies, Denver and Bristol, each own a warehouse, and they agree to an exchange in which no cash changes hands. The following information for the two warehouses is available:...
-
Men Conference Ltd has an authorized share capital of 60,000,000 ordinary shares of Sh. 10 each and 500,000 shares of Sh. 50 each. The following trial balance of the company was extracted as at 31.12...
-
A firm that sells software services has been piloting a new product and has records of 500 customers who have either bought the services or decided not to. The target value is the estimated profit...
-
a. Define the terms: proton, neutron and electron. b. An atom X has a proton number of 19 and relative atomic mass of 39. (i) How many electrons, protons and neutrons are there in an atom of X? (ii)...
-
a. How many atoms of the different elements are there in the formulae of the compounds given below? (i) Nitric acid, HNO 3 (ii) Methane, CH 4 (iii) Copper nitrate, Cu(NO 3 ) 2 (iv) Ethanoic acid, CH...
-
Consider the following two plots: (a) Which of the two plots represents an inverse relationship? Express the inverse relationship in an English statement. (b) A student looks at the plot and declares...
-
Define T P2R2 by T(p) p(0)+: = = P(2) (1) (5 points) If p(t)=1+t+t2, find T(p); (2) (5 points) Is T a linear transformation? Answer Yes or No; (3) (5 points) Find the range of T.
-
A wave travels with speed 1 9 5 m / s. Its wave number is 1 . 4 0 rad / m What is its wavelength and what is its frequency?
-
In a dart gun, a spring with k = 400.0 N/m is compressed 5.1 m when the dart 4.62 kg is loaded. What is the muzzle speed of the dart when the spring is released? Ignore friction. Dart gun (a) before...
-
A standard gold bar stored at Fort Knox, Kentucky, is 7.00 inches long, 3.63 inches wide, and 1.75 inches tall. Gold has a density of 19,300 kg/m3. What is the mass of such a gold bar?
-
Brian needs to make a total of 20 deliveries this week. So far he has completed 14 of them. What percentage of his total deliveries has Brian completed?
-
(a) Write out a pseudo-code algorithm, using both row and column pointers, for Gaussian Elimination with Full Pivoting. (b) Implement your code on a computer, and try it on the systems in Exercise...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Charles's law is sometimes expressed in the form V = Vo (l + a), where is the Celsius temperature, a is a constant, and Vo is the volume of the sample at Oe. The following values for a have been...
-
A constant-volume perfect gas thermometer indicates a pressure of 6.69 kPa at the triple point temperature of water (273.16 K). (a) What change of pressure indicates a change of 1.00 K at this...
-
Calculate the molar volume of chlorine gas at 350 K and 2.30 atm using (a) The perfect gas law and (b) The van der Waals equation. Use the answer to (a) to calculate a first approximation to the...
-
Why would a financial institution's PEP customers be required to provide documentation to evidence their source of wealth and/or income? In other words, what type of fraud would PEP clients more...
-
What is the correct calculation to determine the return rate of the completed questionnaires?
-
What are the Company policies & procedures, any relevant document for Establish an understanding of roles, responsibilities and purpose?

Study smarter with the SolutionInn App


