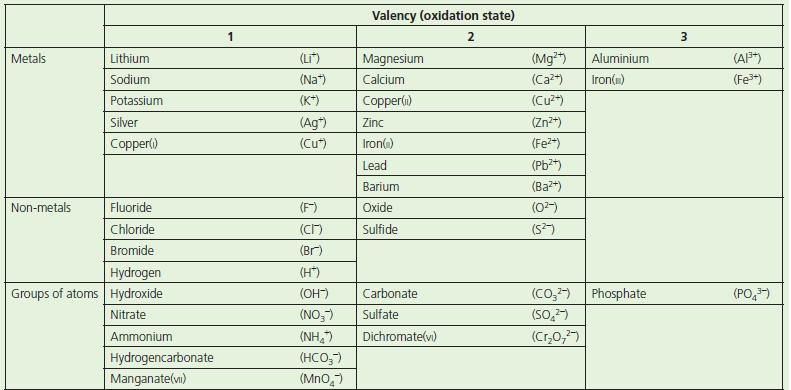
Use the information given in Table 3.6 on p. 43 to work out the formula for: a.
Question:
Use the information given in Table 3.6 on p. 43 to work out the formula for:
a. Silver oxide
b. Zinc chloride
c. Potassium sulfate
d. Calcium nitrate
e. Iron(ii) nitrate
f. Copper(ii) carbonate
g. Iron(iii) hydroxide
h. Aluminium fluoride.
Table 3.6
Valency (oxidation state) 1 Metals Lithium (LI") Magnesium (Mg) Aluminium (AP) Sodium (Na*) Calcium (Ca2") Iron(m) (Fe*) Potassium (K*) Copperi) (Cu2") Silver (Ag") Zinc (Zn") Copperl) (Cu*) Iron() (Fe2*) Lead (Pb) Barium (Ba") Non-metals Fluoride (F) Oxide (02-) Chloride (Cr) Sulfide (S2-) Bromide (Br) Hydrogen (H") Groups of atoms Hydroxide (OH") Carbonate (CO,) Phosphate (PO,) Nitrate (NO,) Sulfate (SO,) Ammonium (NH,") Dichromate(vi) (Cr,0,) Hydrogencarbonate (HCO;) Manganate(vi) (Mno,)
Step by Step Answer:
Compound name a Silver oxide b Zinc Chloride ...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
Use the information given in the previous exercise to determine if there is sufficient evidence to conclude that the mean pulse rate in a dental setting differs from the mean pulse rate in a medical...
-
Use the information given in Problem 2 of Chapter 8, as well as the computer output given here, to answer the following questions about the data from that problem. a. Conduct overall regression F...
-
Use the information given in the following figure to construct a mathematical model for the number of pounds of salt x 1 (t), x 2 (t), and x 3 (t) at time t in tanks A, B, and C, respectively....
-
First Ownership orders merchandise from several suppliers from around the world. Each of the suppliers has different shipping or transportation terms. At the end of December, First Ownership had the...
-
Complete each of the following equations. Then write the Lewis formulas of the reactants and products and identify each reactant as a Lewis acid or a Lewis base. a. AlCl3 + Cl b. I + I2
-
This was the first case involving the limits and powers of government to regulate the Internet to be heard by the U.S. Supreme Court. The American Civil Liberties Union (ACLU) and the federal...
-
How are the elementary magnets aligned in the magnet in Figure P27.3? Data from Figure P27.3
-
Roedel Electronics produces a variety of electrical components, including a remote controller for televisions and a remote controller for DVD players. Each controller consists of three subassemblies...
-
1 Explain briefly about six sigma and capacity maturity models? 2 Explain about the contemporary management practices taking place in Indian business model? 3 4 What is performance management?...
-
The State of Dakota seeks to increase the investment of new business in the state by providing the best wireless communications environment in the country. Dakota has vast land areas suitable for...
-
Atoms of elements X, Y and Z have 16, 17 and 19 electrons, respectively. Atoms of argon have 18 electrons. a. Determine the formulae of the compounds formed by the combination of the atoms of the...
-
The diagram shows the arrangement of the outer electrons only in a molecule of ethanoic acid. a. Name the different elements found in this compound. b. What is the total number of atoms present in...
-
The City of Havisham has a fiscal year ending December 31, Year 5. If the city were to produce financial statements right now, the following figures would be included: -- Governmental activities:...
-
In the movie, The Matrix Reloaded, Neo (80 kg) jumps towards Agent Smith (600 kg) who jumps towards Neo. When they collide, they grab hold of one another. If Neo has a velocity of 25.0 m/s to the...
-
One secondary channel for Apple computers is Best Buy. This channel addition expands Apples market presence. For participation credit, substantively discuss what challenges Apple faces/can face with...
-
Pola Sommers, a massage therapist, needs 5 ounces of a 22% lavender oil solution. She has only 4% and 40% lavender oil solutions available. How many ounces of each should Pola mix to obtain the...
-
Explain why capital and fiat money are not perfect substitutes and discuss what this implies for the rate of return on capital relative to the rate of return on fiat money
-
On one graph show how supply and demand side market failures emerge. How does the market forces clear the failures for normal goods
-
Count the number of arithmetic operations needed to solve a system the "old-fashioned" way, by using elementary row operations of all three types, in the same order as the Gauss-Jordan scheme, to...
-
Find the area of the surface generated by revolving the para- metric curve x = cos 1, y = sin? 1 (0 < I sa/2) about the y-axis.
-
A gas mixture consists of 320 mg of methane, 175 mg of argon, and 225 mg of neon. The partial pressure of neon at 300 K is 8.87 kPa. Calculate (a) The volume and (b) The total pressure of the mixture.
-
In an experiment to measure the molar mass of a gas, 250 cm3 of the gas was confined in a glass vessel. The pressure was 152 Torr at 298 K and, after correcting for buoyancy effects, the mass of the...
-
A certain sample of a gas has a volume of20.00 dm ' at OCand 1.000 atm. A plot of the experimental data of its volume against the Celsius temperature, , at constant p, gives a straight line of slope...
-
GBC Electronics operates a business in Ontario and had the following transactions in the month of December of 2018. The HST in Ontario is 13% and the company uses a perpetual inventory system. Dec 2...
-
Flight Ltd reported a $375,000 loss on the sale of its Eastern Division, net of tax. If Flight's tax rate is 25%, how much was Flight's actual loss? S
-
Daisy Corporation, a calendar year accrual method taxpayer, manufactures and sells life-like plastic flowers. A. Daisy is sued by a customer, Catherine, who is injured in 2024 when she slips and...

Study smarter with the SolutionInn App


