Prove each of these identities. a. cosx tanx sinx b. c. d. e. f. cos 4
Question:
Prove each of these identities.
a. cosx tanx ≡ sinx
b.
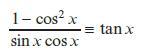
c.
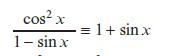
d.
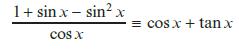
e.
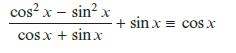
f. cos4 x + sin2 x cos2 x ≡ cos2 x
Transcribed Image Text:
1- cos²x sin x cos x = tan x
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a Proof cos x tan x cos x sin x cos x sin x Therefore cos x tan x sin x b Proof 1 cos ...View the full answer

Answered By

Praveen Mishra
My goal is to help people by providing students with accurate and helpful information that can help them in their studies.
My ability to answer questions and explain has made me a valuable resource for many individuals and organizations.
I have been working as an online tutor for the past six months.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Mathematics Pure Mathematics 1 Coursebook
ISBN: 9781108407144
1st Edition
Authors: Sue Pemberton, Julian Gilbey
Question Posted:
Students also viewed these Mathematics questions
-
Prove that each of these identities is true. You may use any of the identities that have been proved in this chapter. a. secA - sin A tan A = cos A b. 1/sin2A - 1/tan2A = 1 c. secAcosB - tanAsin -...
-
Prove each of these identities. a. cos 2 x sin 2 x = 2 cos 2 x 1 b. cos 2 x sin 2 x = 1 2 sin 2 x c. cos 4 x + sin 2 x cos 2 x = cos 2 x d. 2(1 + cos x) (1 + cos x) 2 = sin 2 x e. 2 (sin x +...
-
Prove each of these identities. a. cos 2 x sin 2 x 2 cos 2 x 1 b. cos 2 x sin 2 x 1 2 sin 2 x c. tan 2 x sin 2 x tan2 x sin 2 x d. cos 4 x + sin 2 x sin4 x + cos 2 x
-
Arginine, the most basic of the 20 common amino acids, contains a guanidino functional group in its side chain. Explain, using resonance structures to show how the protonated guanidino group...
-
How should a company decide which risks to hold and which to hedge?
-
A customer sued a pharmacist and the drug store that employed the pharmacist (Walgreens). The pharmacist reviewed the prescription records of a customer (during her shift at work), learned that the...
-
The time to prepare a slide for high-throughput genomics is a Poisson process with a mean of two hours per slide. What is the probability that 10 slides require more than 25 hours to prepare?
-
Jill Motta, president of Motta Company, has instructed Linda Berger, the head of the accounting department for Motta Company, to report the companys land in the companys accounting reports at its...
-
Question 10 of 11 < > -/3 E A Here are comparative financial statement data for Blossom Company and Crane Company, two competitors. All data are as of December 31, 2022, and December 31, 2021....
-
Schweizer Distribution specializes in distributing fresh produce to local restaurants in the Chicago area. The company currently sells 12 different products through the efforts of three sales...
-
Draw a diagram showing the quadrant in which the rotating line OP lies for each of the following angles. On each diagram, indicate clearly the direction of rotation and state the acute angle that the...
-
Find the value of x satisfying the equation sin 1 (x 1) = tan 1 (3).
-
Indicate several ways in which the valence bond method is superior to Lewis structures in describing covalent bonds.
-
Consider a nonviscous fluid undergoing laminar flow in an open, horizontal channel. The Bernoulli effect says that where the flow speed is greater, the pressure is smaller, but the pressure at the...
-
Apple wanted to compete more effectively with Amazons Kindle so more iPad owners would read books on iPads instead of Kindle. Apple met with the six largest book publishers and promised them better...
-
The capped end of a pipe in your basement springs a leak. The pipe has a diameter of \(50.0 \mathrm{~mm}\) and is attached to a joist so that it is \(2.00 \mathrm{~m}\) above the basement floor. The...
-
Wyndham Hotels website claimed to protect customers information. Hackers cracked Wyndhams customer base three times in a year, costing the customers at least $10 million. The FTC sued claiming it was...
-
A drop of liquid is placed on a surface. A particle of the liquid at the contact point is subject to a cohesive force of \(4.00 \mathrm{~N}\) directed \(52.0^{\circ}\) above the surface. If the...
-
For each of the following, write the molecular equation, including phase labels. Then write the net ionic equation. The salts formed in these reactions are soluble. Discuss.
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Answer Problem 4.15 for a mixed suburban/rural hospital (hospital E). Infectious Disease Newborns were screened for human immunodeficiency virus (HIV) or acquired immunodeficiency syndrome (AIDS) in...
-
Answer Problem 4.16 for a mixed suburban/rural hospital (hospital E). Infectious Disease Newborns were screened for human immunodeficiency virus (HIV) or acquired immunodeficiency syndrome (AIDS) in...
-
Suppose 10 gonorrhea cases are reported over a 3-month period among 10,000 people living in an urban county. The statewide incidence of gonorrhea is 50 per 100,000 over a 3-month period. Is the...
-
The following information is available for Tamarisk Company. Raw materials inventory Work in process inventory Materials purchased in April $94,400 Direct labor in April 76,800 Manufacturing overhead...
-
Your basketball team has two coaches, Coach Mike and Coach Brian. Coach Mike decides the lineup for 60% of the games, and Coach Brian decides the lineup for 40% of the time. You love playing point...
-
Personal Reflective report on Coaching and Mentoring what is coaching and mentoring? own experience of coaching and mentoring Any skills you might have in coaching Discuss how you can utilize this...

Study smarter with the SolutionInn App


