A well-insulated 1.2 kg block of iron is heated using a 50 W heater for 4.0 min.
Question:
A well-insulated 1.2 kg block of iron is heated using a 50 W heater for 4.0 min. The temperature of the block rises from 22 °C to 45 °C. Find the experimental value for the specific heat capacity of iron.
You will need to use data from Table 19.3 to answer these questions.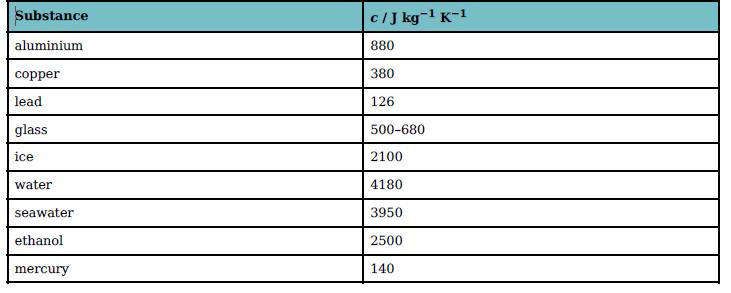
Substance cIJ kg-1 K-1 aluminium 880 copper 380 lead 126 glass 500-680 ice 2100 water 4180 seawater 3950 ethanol 2500 mercury 140
Step by Step Answer:
Given A well insulated block of iron has Massm 12kg which is heated with the help of a heater hav...View the full answer



Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
A large, cold (0.0C) block of iron is immersed in a tub of hot (100.0C) water. In the first 10.0 s, 41.86 kJ of heat are transferred, although the temperatures of the water and the iron do not change...
-
For this assessment, you will need to use the SAS Enterprise Guide or the Excel Calculators, the latter of which can be found in Resources. SAS users can reference the SAS tutorials located in the...
-
A 3 kg block of iron at 800oC is dropped into 50 kg of water in an insulated cooling tank. If the final temperature (T2) at equilibrium is 29.9oC, determine (a) The change in internal energy (U). (b)...
-
Discuss the differences between @classmethod , @staticmethod , and instance methods in Python.
-
Mei Li invested $350 at the end of each quarter at 3.2% compounded quarterly. At the end of five years, she was able to withdraw equal amounts at the end of each quarter for nine years. How much is...
-
How might the project manager integrate the various control tools into a project control system?
-
Choose an appropriate closed system and draw a bar diagram representing the energy conversions and transfers that occur during each process of Checkpoint 7.9: (a) a ball launching as the compressed...
-
Gilbert Church owned Church Farms, Inc., in Manteno, Illinois. Church advertised its well-bred stallion Imperial Guard for breeding rights at $50,000, directing all inquiries to Herb Bagley, Manager....
-
Duncan's Diamond Bit Drilling Corporation (Duncan) purchased the following assets in 2023. Assume its taxable income was $60,000 for purposes of computing the 179 expense deduction. Asset Purchase...
-
Go through the transaction list (Document1) and identify which cycle each transaction belongs to. A transaction will belong to only one cycle; either the Sale to Cash Receipt (S/CR) Cycle, Purchases...
-
a. A 500 W kettle contains 300 g of water at 20 C. Calculate the minimum time it would take to raise the temperature of the water to boiling point. b. The kettle is allowed to boil for 2 minutes....
-
The resistance of a thermistor at C is 2000 . At 100 C the resistance falls to 200 . When the thermistor is placed in water of constant temperature, its resistance is 620 . a. Assuming that the...
-
Refer to the opening feature in this chapter about Michael Chasen and Matthew Pittinsky and their company, Blackboard. Assume that they must acquire the Japanese rights to certain educational...
-
What is the difference between an Article III and an Article I court?
-
What can the 3-way match detect?
-
Looking for a few questions that are necessary for understanding justice are? why are thes questions needed to understand the importance of justice and the keywords associated with this topic? need...
-
What role do the generally accepted auditing standards ( GAAS ) play in the audit function?
-
Tinsel Inc. purchased equipment on January 1, 20X1 for $15,000 and immediately sold it to it's parent, Pretzel Inc. for $20,000. The equipment is expected to have a remaining useful life of five...
-
Electrons in a cathode ray tube start from rest and are accelerated through a potential difference of 12.0 kV. They are moving in the + x -direction when they enter the space between the plates of a...
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
When ethoxybenzene is treated with a mixture of nitric acid and sulfuric acid, two products are obtained each of which has the molecular formula C 8 H 9 NO 3 . (a) Draw the structure of each product....
-
The pressure dependence of G is quite different for gases and condensed phases. Calculate G m for the processes (C, solid, graphite, 1 bar, 298.15 K) (C, solid, graphite, 325 bar, 298.15 K) and (He,...
-
Many biological macromolecules undergo a transition called denaturation. Denaturation is a process whereby a structured, biologically active molecule, called the native form, unfolds or becomes...
-
What is expected price of the bond in 4 yrs? Assume the interest rate will remain the same. par = 1000 Yrs to maturity = 25 Coupon = 8% paid semi-annually B= 1.5 Rf = 4% Market Risk Premium = 8%
-
How many pints of milk must be purchased for 148 servings, if each person is to receive 4.8 ounce glass? show your solution
-
Does the concept of revenue less expense equaling an increase in equity or fund balance make sense to you? If not, why not?

Study smarter with the SolutionInn App


