What is the voltage across the 3.0 k resistor in Figure 11.9 when the light intensity is
Question:
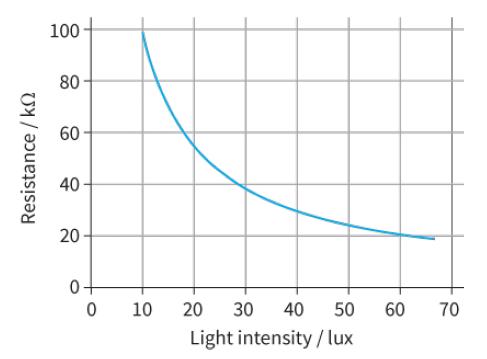
What is the voltage across the 3.0 kΩ resistor in Figure 11.9 when the light intensity is 10 lux?
Transcribed Image Text:
100 80 60 40 20 0+ 10 20 30 40 50 60 70 Light intensity/lux Resistance / k
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Using the concept of potential divider When R 20 ...View the full answer

Answered By

KARTIKAY SRIVASTAVA
#CBSE class 10th - 10 CGPA , 100/100 in MATHEMATICS.
#CBSE class 12th - 10 CGPA , 99/100 in MATHEMATICS.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
What is the voltage across six 1.5-V batteries when they are connected (a) In series, (b) In parallel, (c) Three in parallel with one another and this combination wired in series with the remaining...
-
What is the voltage of a voltaic cell based on this reaction? Is the reaction spontaneous as written? 5Ni + 2MnO4 + 16H+ ( 3Mn2+ + 8H2O + 5Ni2+
-
What is the voltage drop across RL? A. 0.22 V B. 0.33 V C. 0.75 V D. 1.50 V The diagram shows a small water heater that uses an electric current to supply energy to heat water. A heating element, RL,...
-
How do patterns of mental illness differ according to population? Name examples of where one psychological symptom could be interpreted in two different ways based on age/gender/identified problem.
-
The amount of $27 350 is invested at 6% compounded monthly for 6 years. After the initial 6-year period, the balance in the fund is converted into an annuity due paying $1600 every 6 months. If...
-
Castle Lager has just purchased the Jacksonville Brewery. The brewery is two years old and uses absorption costing. It will sell its product to Castle Lager at $ 47 per barrel. Peter Bryant, Castle...
-
Draw and determine the C.M. positions of the following molecules. Use a periodic table of the elements for the masses of the elements (it is enough to consider the mass number). Atomic distances are...
-
Kickapoo Company uses an imprest petty cash system. The fund was established on March 1 with a balance of $100. During March, the following petty cash receipts were found in the petty cash box. The...
-
Explain why clients may want to have a review, versus an audit. In detail, explain the differences between a certified audit and a review. In addition, what role does an internal auditor play in...
-
1. Use descriptive statistics to summarize the data collected by Fresnos Manager of Product Quality for each of the 5 randomly selected dice. Based on these descriptive statistics, what are your...
-
This circuit shows a potential divider. The battery has negligible internal resistance and the voltmeter has infinite resistance. a. State and explain how the reading on the voltmeter will change...
-
This diagram shows two circuits that could be used to act as a dimmer switch for a lamp. a. Explain one advantage circuit 1 has over circuit 2. b i. The lamp is rated at 60 W at 240 V. Calculate the...
-
Use Exercise 51 to find (lnx)3dx. In Exercise 51 (ln x)n dx = x(ln x)n - n (ln x)n-1 dx
-
How do chromatin organization and histone modifications influence nuclear processes such as transcription and DNA replication?
-
Factor to solve the following equation for x. (SHOW YOUR WORK) 923 +272-2x-6=0
-
3 a a. [2] Does Prim's algorithm always work correctly on graphs with negative edge weights? b. [2] Apply Prim's algorithm on the following graph. Show the construction of the minimum spanning tree...
-
2 (s)' Solid lead(II) hydroxide, Pb(OH), in a test tube will dissolve when excess 6 M NaOH is added. Solid iron(II) hydroxide, Fe(OH), however, does not dissolve when excess 6 M NaOH is added. Answer...
-
Current Attempt in Progress The Ivanhoe, Inc. sold 10,350 season tickets at $2,040 each. By December 31, 2025, 16 of the 40 home games had been played. What amount should be reported as a current...
-
A square loop of wire of side 2.3 cm and electrical resistance 79 is near a long straight wire that carries a current of 6.8 A in the direction indicated. The long wire and loop both lie in the...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
One liter of fully oxygenated blood can carry 0.18 liters of O 2 measured at T = 298 K and P = 1.00 atm. Calculate the number of moles of O 2 carried per liter of blood. Hemoglobin, the oxygen...
-
Determine whether the pinacol rearrangement, shown in the previous problem, is a reduction, an oxidation, or neither. Resonance stabilized H,SO, eat
-
discuss the concept of risk-adjusted profit margins, elucidating methodologies for incorporating risk management strategies into financial planning processes ?
-
The radiology department at St. Joseph's Hospital, a not-for-profit, is considering purchasing a magnetic resonance imaging (MRI) machine. The cost to purchase and install an MRI is approximately...
-
For the following models, determine whether they are stationary or not. If yes, calculate their autocovariance functions (ACFs), that is, Yo, Y1, V2, .. (a) DGP1: yt = 0.75yt-1+ Et (b) DGP2: Yt =...

Study smarter with the SolutionInn App


