a. Draw the skeletal formula of pentane, a straight chain hydrocarbon with a molecular formula of C
Question:
a. Draw the skeletal formula of pentane, a straight chain hydrocarbon with a molecular formula of C5H12.
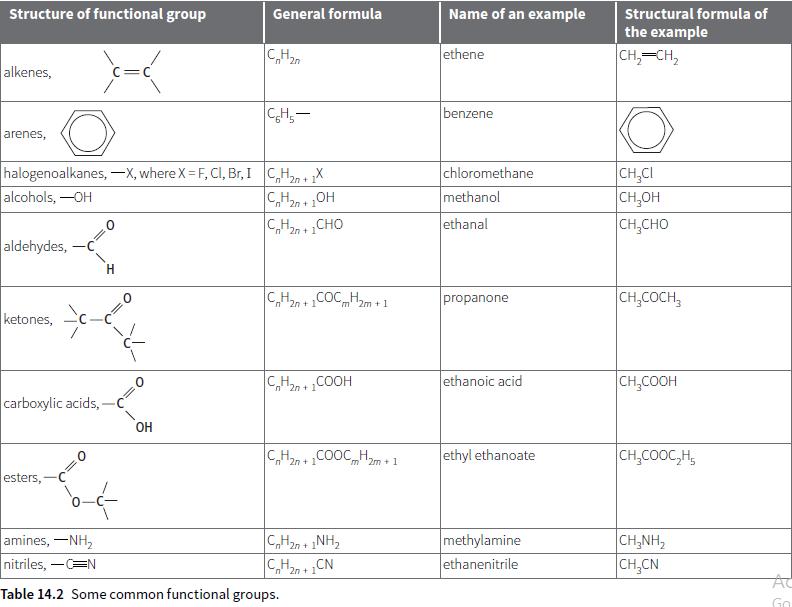
b. Draw the structural formulae of the molecules shown in Figure 14.2, parts d, e and f.
Transcribed Image Text:
Structure of functional group General formula Name of an example Structural formula of the example C,H2 ethene CH,-CH, n "2n alkenes, C=C CH,- benzene arenes, halogenoalkanes, –X, where X = F, CI, Br, I C,H, + X alcohols, -OH chloromethane methanol CH,CI CH,OH CH,CHO "2n 2n+ C,H2n + 1CHO ethanal aldehydes, C,H2n +COCHm + 1 CH,COCH, propanone "m' ketones, C,H2n + COOH ethanoic acid CH,COOH carboxylic acids, OH C,Han +COOC Hm+1 ethyl ethanoate CH,COOC,H, m' 2m + 1 esters,- amines, -NH, nitriles, -CN methylamine ethanenitrile CH,NH, 2n + C,H20 + CN CH,CN AC Table 14.2 Some common functional groups. Go
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
a Draw the skeletal formulae of: i. Pentan-2-one ii. Pentan-3-one iii. Pentanal. b. Describe the results you would expect to see if pentan-3-one and pentanal were separately treated with Tollens...
-
a. Draw the displayed formula and the skeletal formula of cyclopentane. b. What is the general formula of cycloalkanes? c. Give two differences between a molecule of cyclopentane and a molecule of...
-
Draw two different structural formulas based on the molecular formula C2H6O. Is the fact that you can have more than one compound with the same molecular formula consistent with Dalton's atomic...
-
XY is a pharmaceutical company with the head-quarter in the United States. However, its cash flow depends only on sales in Japan. It receives a cash flow of Yen 20 billion with certainty in t=1. The...
-
This problem is based on information taken from The Rating Guide to Life in Americas Fifty States, by G. S. Thomas. A random sample of n1 = 288 voters registered in the state of California showed...
-
Metters Cabinets, Inc., needs to choose a production method for its new office shelf, the Maxistand. To help accomplish this, the firm has gathered the following production cost data: Metters...
-
Boswell manufactures high-quality speakers. Suppose Boswell is considering spending the following amounts on a new quality program: Boswell expects this quality program to save costs as follows: It...
-
Sportway Inc. produces high-quality tennis racquets and golf clubs using a patented forming process and high-quality hand-finishing. Products move through two production departments: Forming and...
-
Total Energy (a) Conserved 12. Which one of the following is true in the case of inelastic collisions? Kinetic Energy Conserved Momentum Conserved (b) Conserved Not Conserved Conserved (c) Conserved...
-
Construct a graph that can be used for sensitivity analysis for the preceding problem. a. How sensitive is the solution to the problem in terms of the probability estimate of .35? b. Suppose that,...
-
A chemist was investigating the best way to produce 1,2-dichloroethane. He devised two methods, I and II, of doing this. I . He reacted ethane with chlorine in the presence of UV light by the...
-
Draw a 3D formula for: a. Propane b. Propene.
-
I still dont get it, said Ahmir, how can all the monthly loan repayments we make to the bank for the dining room expansion project we completed last year not be considered an operating expense? We...
-
In the state of Florida, the intestate succession statute elects an estate executor through probate court, if there isn't a will in place. The next of kin is selected. The next of kin starts with a...
-
You just won the "NoLuck" lottery, and you selected the one lump sum option. You plan to use it for 25 years and make quarterly withdrawals of $50,000. The APR is S percent a year, compounded...
-
A paper mill, P, competes in a perfectly competitive international market. P produces 10 tons of paper daily. The market price = $100 a ton. For every ton of production, P produces 0.5 tons of...
-
(a) A blanket with a shiny reflective coating on the inside will help you stay warmer at night than a regular blanket. Why? What principles and equations might be involved? [10 points] (b) A spinning...
-
Evaluate If n is an even integer, then n - 1 is odd.
-
Explain how Carnival Corporation accounts for customer deposits as passengers purchase cruise tickets in advance.
-
For the following exercises, find the inverse of the function and graph both the function and its inverse. f(x) = 4 x 2 , x 0
-
A vessel containing a liquid is opened inside an evacuated chamber. Will you see a liquidgas interface if the volume of the initially evacuated chamber is a. Less than the critical volume, b. A...
-
Calculate S R for the reaction H 2 (g) + Cl 2 (g) 2HCl (g) at 870. K. Omit terms in the temperature dependent heat capacities higher than T 2 /K 2 .
-
Use the result of Problem P3.10 to derive a formula for (CV /V ) T for a gas that obeys the RedlichKwong equation of state, RT 1 a Vm - b VT VVm + b)' T VVm P:
-
A truck driver averages 92 kilometers per hour between 2 p.m. and 6 p.m. Can you determine the speed of the truck at 4 p.m.?
-
The Hernys law contant for nitrogen dioxide is k H = 4.0x10 -2 M/atm at 25C. What is the solubility of NO 2 at 25C in water at a partial pressure of 575 mmHg?
-
A project costs Birr 2,000,000 and yields annually a profit of Birr 300,000 after depreciation at 12.5% but before tax at 50%. Calculate the payback period.

Study smarter with the SolutionInn App



