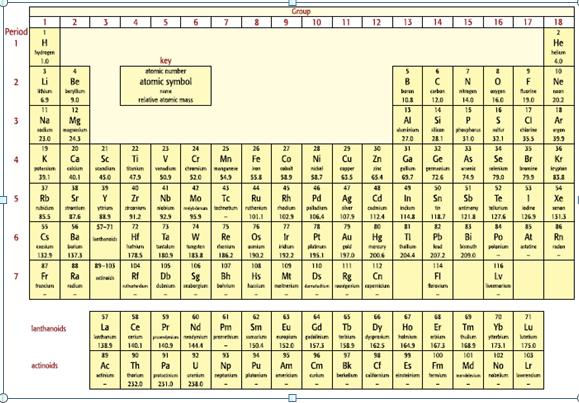
Look at the elements in Period 2 of the Periodic TABLE. Using your knowledge of Period 3
Question:
Look at the elements in Period 2 of the Periodic TABLE. Using your knowledge of Period 3 elements, predict and explain the relative sizes of:
a. The atomic radii of lithium and fluorine.
b. A lithium atom and its ion, Li+.
c. An oxygen atom and its ion, O2−.
d. A nitride ion, N3−, and a fluoride ion, F−.
Transcribed Image Text:
2 3T4 10 11 12 13 14 15 16 17 18 Period key atomi umber 2 atomic elative atmic mas 3 4 6 lanthanoids Nd 1625 actinoids Th Np tharium pian cteium 232.0 2313 234.0 中 に . トz1 ala】 a2是 ||el. ト81a | |a a8|出】, a品| 1+p |よ =1コ12 aao
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a The atomic radii of lithium Li and fluorine F are different due to the number of electrons and the ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Group 2 of the periodic table contains the family of elements called the alkaline earths. How active chemically would you expect an alkaline earth element to be compared with the alkali metal next to...
-
The nitride ion and the amide ion, NH2, have greater attractions for the hydronium ion than the hydroxide ion does. Write the equations for the reactions that occur when calcium nitride and sodium...
-
Atomic lithium of concentration n = 3.6.1016 cm 3 is at a temperature T = 1500 K. In this case the power emitted at the resonant line's wavelength = 671 nm (2P 2S) per unit volume of gas is equal to...
-
Consider a long cylindrical solenoid with diameter R, number of current loops N and length L through which a current I runs. Now (a) Use Ampre's law to calculate the magnetic field inside the...
-
The following table shows ceremonial ranking and type of pottery sherd for a random sample of 434 sherds at a location in the Sand Canyon Archaeological Project, Colorado (The Architecture of Social...
-
How is the variance of the total project computed in PERT?
-
Read the blog post We Will Never See Anything Like It Again: Movements in Real GDP during the Covid19 Recession at...
-
JIT production, relevant benefits, relevant costs, ethics. Parson Container Corporation is considering implementing a JIT production system. The new system would reduce current average inventory...
-
11.A simple LR circuit is connected to a battery at t = 0. The time instant at which rate of energy storage in inductor is half of power delivered by battery 2L (1) In 2 (3) In 2 (2) In (4) (4) In 3
-
G. Menges developed the following econometric model for the West German economy: where Y = national income I = net capital formation C = personal consumption Q = profits P = cost of living index R =...
-
a. Explain what is meant by the term periodic property. b. The graph shows how a periodic property varies when plotted against atomic number for Period 3 (sodium to argon). i. Identify the property....
-
The variation of melting point with atomic number for Periods 2 and 3 is shown in the graph below. a. Explain what we mean when we say melting point is a periodic property. b. Explain the following....
-
Refer to the information in BE65. Calculate ending inventory and cost of goods sold for the year, assuming the company uses LIFO. Data from in BE6-5 During the year, Wright Company sells 470...
-
The logical fallacy that has affect me in my past and my plan to correct this issue is the informal fallacies. This fallacy has led me to started arguments or have debates with many of my friends in...
-
Provide a brief but clear explanation 1)The rapid expansion of middle class is likely to aid not only the growth of the Asian economy but also the global economy. 2)Real effective exchange rate and...
-
Why is Cadre so concerned (more so than the typical real estate investment firm) about selecting profitable deals? How quickly do you think Cadre should launch their secondary market? Why? How...
-
What are some other techniques that can be used to hide malicious activity on a Windows or a Linux/Unix system?
-
What are three objectives of an OS design? 2. What is the kernel of an OS? 3. What is multiprogramming? 4. What is a process? 5. List and briefly explain five storage management responsibilities of a...
-
The pharmaceutical industry spends billions of dollars each year on research and development. Rather than capitalize these R&D expenditures as intangible assets , companies are required to charge...
-
Access the Federation of Tax Administrators Internet site at www. taxadmin.org/state-tax-forms and indicate the titles of the following state tax forms and publications: a. Minnesota Form M-100 b....
-
Predict which side of the following equilibrium is favored, and explain your choice. Heat
-
When trans-3,4-dimethylcyclobutene is heated, conrotatory ring opening can produce two different products, yet only one is formed. Draw both products, identify which product is formed, and then...
-
Predict the product for each of the following reactions: (a) (b) (c) Heat Heat
-
Suppose you model the relationship between used car prices (measured in dollars) and mileage (measured in miles) and estimate the relationship by OLS in STATA. The result is as follows: Source SS df...
-
Chicken Consumption In 2001 Americans ate, on average, 56 pounds of chicken annually. This amount is expected to increase to 61 pounds in 2012. (Source: Department of Agriculture.) (a) Determine a...
-
Jessie Jonny is the director of Biryani Limited. While acting for the company, he becomes aware of a business opportunity and acquires a huge debt for doing the business by mortgaging the factory...

Study smarter with the SolutionInn App


